POCUS Spotlight: Ultrasound-Guided Point of Care Diaphragm Assessment
Cite as: Kwofie K, Kalagara H, Girón-Arango L. Ultrasound-guided point of care diaphragm assessment. ASRA Pain Medicine News 2025;50. https://doi.org/10.52211/asra080125.010.
POCUS Spotlight
Introduction
The first description of ultrasound-guided diaphragmatic assessment was in 1969.1 Still, more recently, due to its reproducibility and reliability, it has become a modality of choice for assessing and monitoring diaphragm function.2 Moreover, it can be used alongside the routine lung ultrasound examination, which was described previously in this newsletter, providing further information to the clinician.3
The three most common techniques used to assess diaphragmatic function with ultrasound are the dome of the diaphragm (DOD) excursion analysis, zone of apposition (ZOA) thickening analysis, and the ZOA excursion analysis. This review will discuss the diagnostic decision-making around these techniques to compare and contrast their utility.
Background/Anatomy
The diaphragm is a thin, musculotendinous, domed-shaped structure that separates the thoracic cavity from the abdomen. It is the major muscle of respiration and shields the thorax from intrabdominal pressures. It is innervated by the phrenic nerve, which arises from the ventral rami of C3 through C5. The diaphragm has three segments: the central tendon, the costal diaphragm, and the crural diaphragm. The central tendon is non-contractile, allowing blood vessels to pass between the thoracic and abdominal cavities. The costal diaphragm connects the ribs and the xiphoid to the central tendon. The crural diaphragm connects the central tendon to the first three lumbar vertebrae. The left hemidiaphragm typically lies 1.5-2.5 cm below the right hemidiaphragm.4 As the muscle contracts and shortens, it causes the diaphragm to be displaced caudally, causing a reduction in the intrapleural pressure and an increase in intraabdominal pressure. This results in inward movement of gases into the lungs, provided that the airway is patent, to provide normal, negative pressure, physiologic inhalation (see Figure 1).
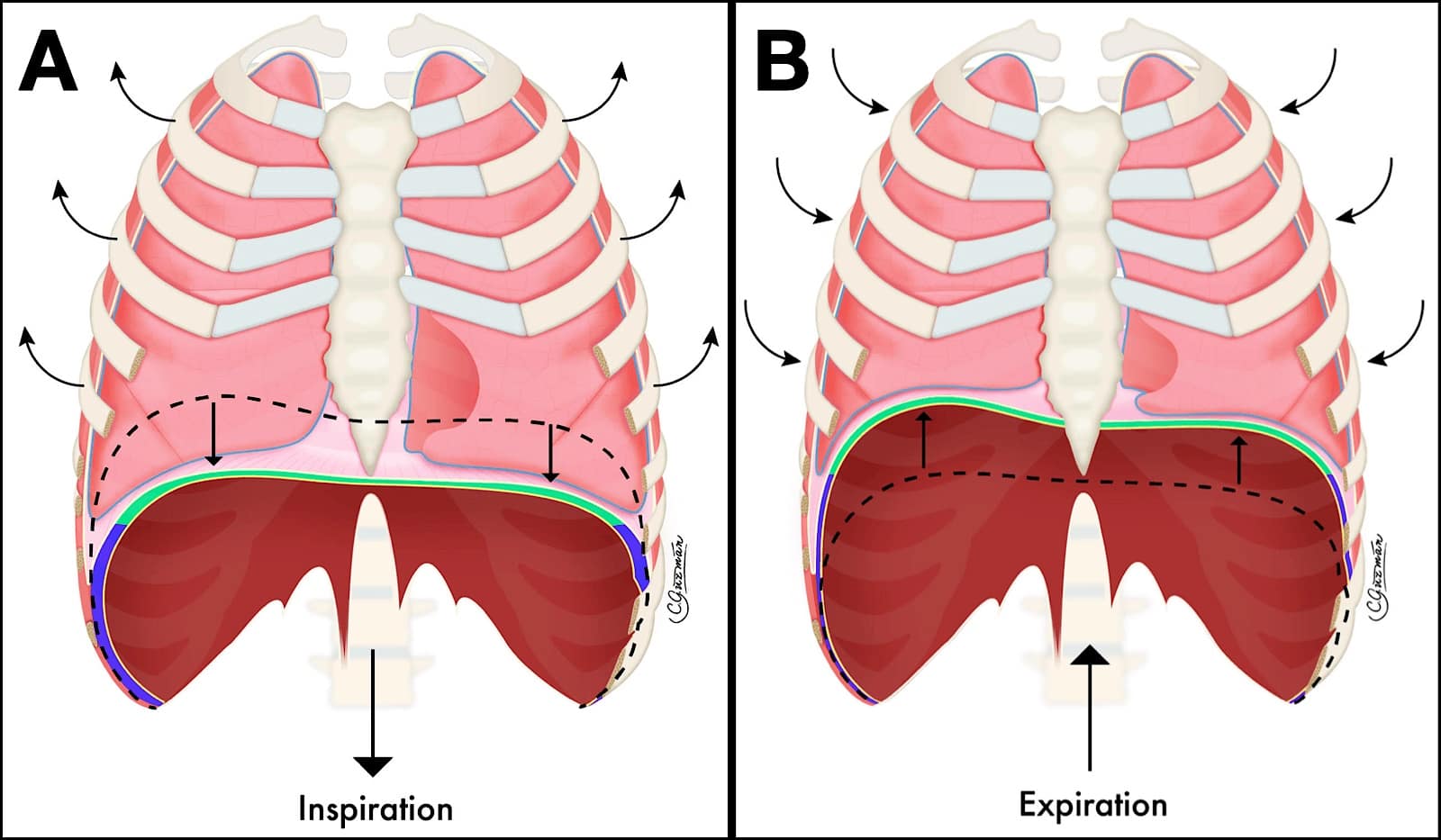
Reproduced with permission from Giron Arango Medicine Professional Corporation.
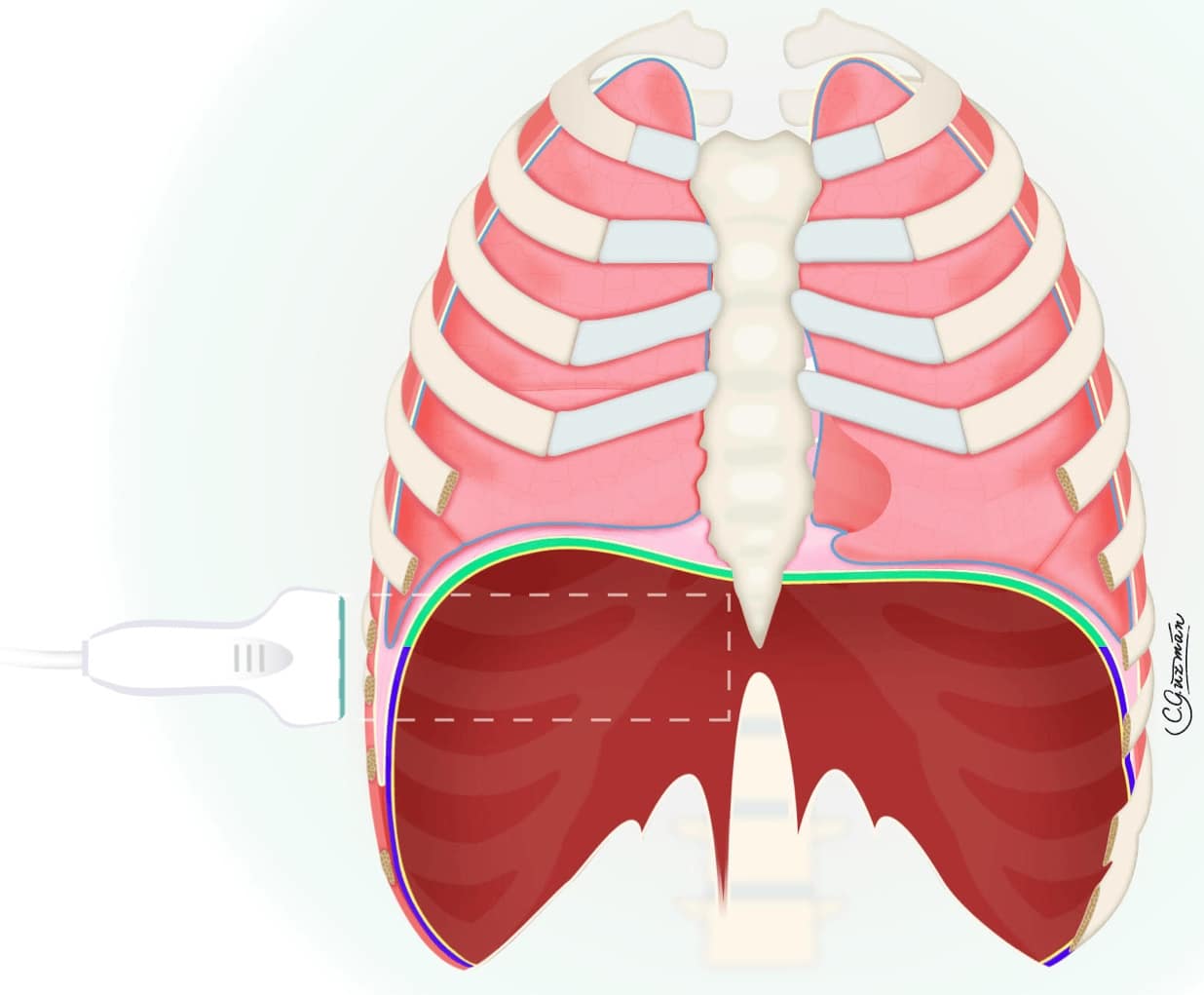
From a functional point of view, each hemidiaphragm can be divided in two areas: the DOD, which is composed primarily by the central tendon and serves to separate the lung from the abdomen, and the ZOA. The ZOA is the costophrenic recess where the costal diaphragm is in direct contact with the internal rib cage without any lung tissue (See Figure 2). Unilateral diaphragmatic paresis is frequently asymptomatic in healthy adults. However, dysfunction of the diaphragm can lead to prolonged ventilation in critically ill patients and failed extubation in surgical patients.
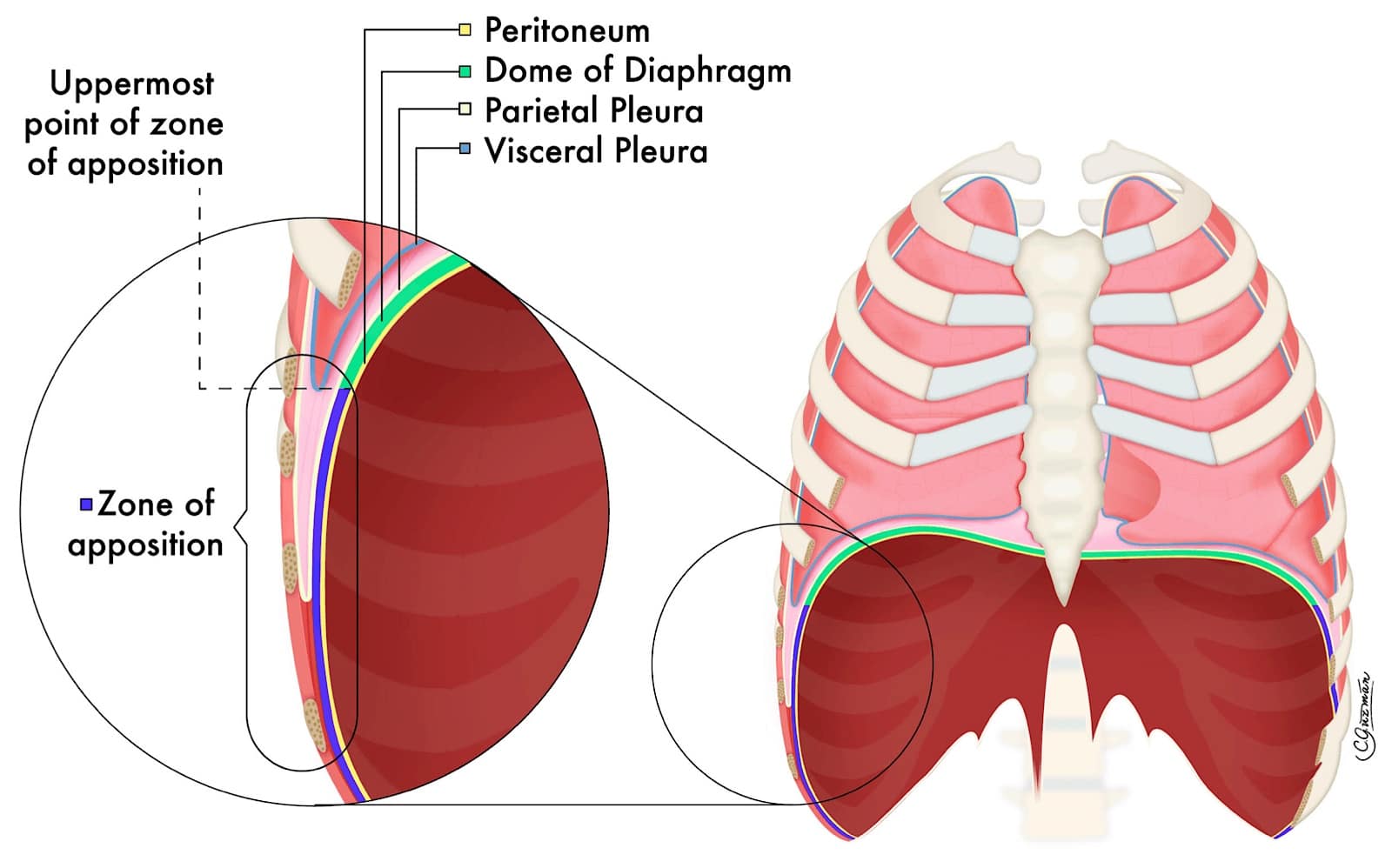
Reproduced with permission from Giron Arango Medicine Professional Corporation.
Diaphragmatic paresis can be diagnosed by chest radiograph, dynamic fluoroscopy, transdiaphragmatic pressure measurement, phrenic nerve stimulation (gold standard), and dynamic magnetic resonance imaging.2,5,6 While a chest X-ray may raise suspicion for a diaphragmatic paresis, the positive predictive value is only 33%.7 However, ultrasound has emerged as a routine assessment tool for diaphragmatic function for anesthesiologists8 due to its safety (non-invasive), reliability, portability, availability, and lack of ionizing radiation. It also allows for real-time, repeatable monitoring to assess and reassess the patient readily. Ultrasound of the diaphragm has been used to monitor recovery of diaphragmatic function,9 to assess for phrenic nerve injury after cardiothoracic surgeries,10,11 and to assess phrenic nerve function and presence of hemidiaphragmatic dysfunction before and after a brachial plexus block, particularly above the clavicle.12-14
Indications
Clinical findings of patients with diaphragmatic paresis can include respiratory distress, unexplained dyspnea, an unexplained unilateral elevated hemidiaphragm, difficulty weaning from oxygen therapy or mechanical ventilation, or unilateral lung collapse. However, many healthy patients with unilateral diaphragmatic paresis may have no symptoms at all.
A well-known phenomenon to regional anesthesiologists is the risk of unilateral hemidiaphragmatic paresis after brachial plexus block due to the proximity of the phrenic nerve. Medical causes include tumor invasion, spinal cord injury, and upper motor neuron diseases (such as amyotrophic lateral sclerosis or poliomyelitis) and botulism. Many cardiac and thoracic surgeries carry a risk of phrenic paresis from transection, stretch, crush, or hypothermia from ice that is used for cardio-protection.15 Thymectomy, in particular, has a 7% incidence of phrenic nerve injury.8 Causes of diaphragmatic paresis are listed in Table 1.
| Location of the Lesion | Etiology |
| Brain (internal capsule or cerebral cortex) | Vascular accident Arnold-Chiari disease |
| Spinal cord | Traumatic Degenerative (severe spondylosis) |
| Motor neurons | Post-polio syndrome Amyotrophic lateral sclerosis Syringomyelia Paraneoplastic neuropathy associated with antibodies-HU Radiation Spinal muscular atrophy |
| Brachial plexus | Traumatic Proximal brachial plexus block Radiotherapy Idiopathic |
| Phrenic | Trauma Cardiac/thoracic surgeries Mediastinal neoplasms (Compression/infiltration) Aortic aneurysm Guillain-Barré Infection (herpes-zoster virus, pneumonias, lyme disease, HIV) Amyotrophic neuralgia (Parsonage-Turner) Others [malnutrition, diabetes, hypothyroidism, porphyria, vasculitis] Idiopathic |
| Lung | Asthma Chronic obstructive pulmonary disease |
| Neuromuscular junction | Myasthenia gravis Botulism Lambert-Eaton syndrome |
| Muscular | Muscular dystrophies Steroid myopathy Pompe disease Myositis Mechanical ventilation |
Any patient at risk of diaphragmatic paresis, experiencing unexplained respiratory distress, or requiring oxygen therapy may be assessed for diaphragmatic dysfunction if it would impact patient management. However, it is important to acknowledge that the presence of subcutaneous emphysema can limit all sonographic methods.
Ultrasound has emerged as a routine assessment tool for diaphragmatic function for anesthesiologists8 due to its safety (non-invasive), reliability, portability, availability, and lack of ionizing radiation.
Acquisition and Interpretation
As stated previously, the three commonly used methods for assessing diaphragmatic function with ultrasound are the DOD excursion analysis, the ZOA thickening analysis, and the ZOA diaphragm excursion analysis. Each method will be discussed in turn for both acquisition and interpretation.
Dome of The Diaphragm (DOD) Excursion Analysis
For the assessment of the DOD, a subcostal approach is required using the liver and spleen as acoustic windows on the right and left sides, respectively, to facilitate the visualization of the diaphragm. Once visualized, the diaphragm is displaced towards the transducer during inhalation, which will appear as an upward deflection on the ultrasound screen. This method is the preferred one for measuring diaphragmatic excursion with ultrasound using both B and M modes.2 Diaphragmatic velocity can also be calculated using this approach and has been used in the assessment of neurodegenerative disease and critical illness, but this is beyond the scope of this article.2
The patient should be positioned supine to maximize diaphragmatic excursion and exaggerate the paradoxical movement of the diaphragm. The presence of large pleural effusions can cause paradoxical diaphragmatic motion in the upright position, which may also explain why the supine position is preferred.2
A curvilinear or phased array low-frequency transducer (2-6 MHz) is ideal due to the depth that is required (up to 20 cm). An abdominal or cardiac preset is typically appropriate. The transducer indicator is generally positioned laterally. The transducer should be placed between the mid and anterior axillary lines, and it should be directed medial, cephalad, and posterior to image the posterior-medial portion of the diaphragm (see Figure 3). The right diaphragm is measured through the hepatic acoustic window, and the left diaphragm is visualized through the splenic acoustic window. The diaphragm can be recognized as a thin hyperechoic arched line that separates the thoracic cavity from the abdominal cavity. One of the limitations of this method is the challenging visualization of the left hemidiaphragm due to an air-filled stomach and the smaller acoustic window provided by the spleen. Literature has reported failure rates ranging from 50% to 80% for the assessment of the left hemidiaphragm.16,17 To improve visualization on the left side, the posterolateral approach may be preferred, positioning the probe posterior to the midaxillary line and directed cephalad, towards the right shoulder (see Figure 3).2 The goal is to visualize the inner third of the diaphragm, which has the most craniocaudal excursion.18 Hepatomegaly with an enlarged left lobe or splenomegaly can facilitate imaging on the left side.
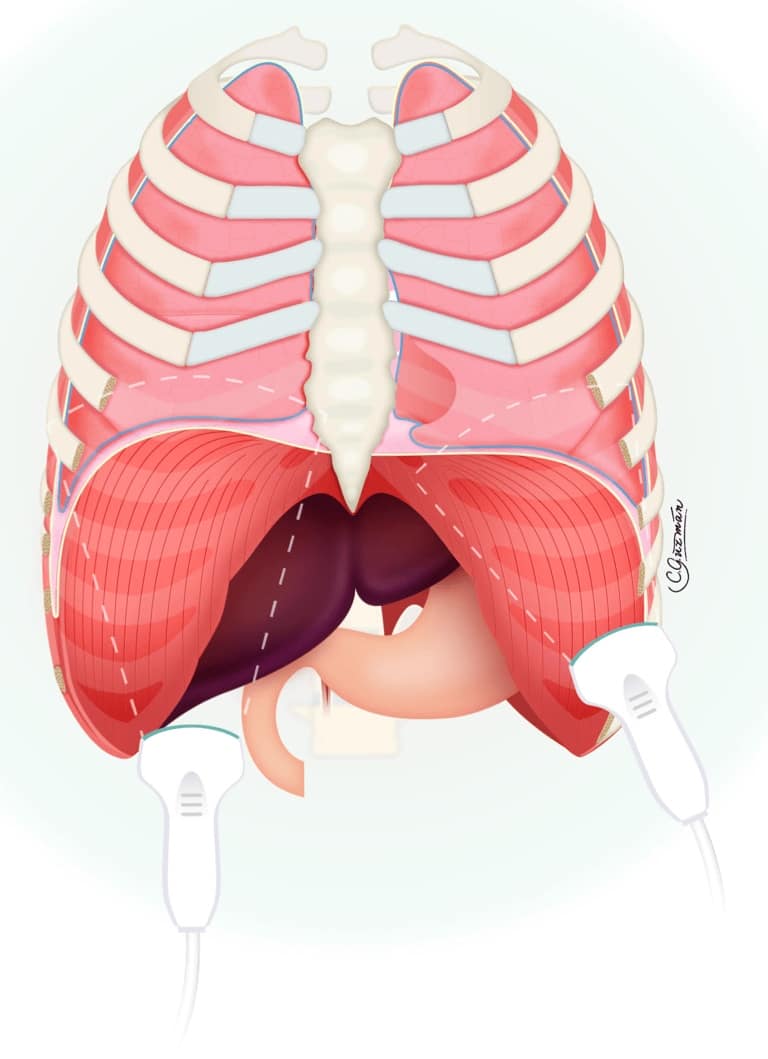
Reproduced with permission from Giron Arango Medicine Professional Corporation.
Diaphragmatic movement is measured at three time points: during quiet breathing, during a maximal inspiratory effort, and during a voluntary sniff (often referred to as a sniff test). In a healthy patient, during quiet breathing minimal excursion will be displayed with an increased caudal motion (towards the transducer) using an M-mode examination, during maximal inhalation or during a sniff test (see Figure 4). In healthy volunteers, normal diaphragmatic excursion ranges from 1 to 2 cm during quiet respiration and 5 to 7 cm during deep breathing.19,20 The lower limit of normal diaphragmatic excursion was 0.9 cm for women and 1 cm for men during quiet breathing, 1.6 cm for women and 1.8 cm for men during a sniff test, and 3.7 cm for women and 4.7 cm for men during deep breathing.17
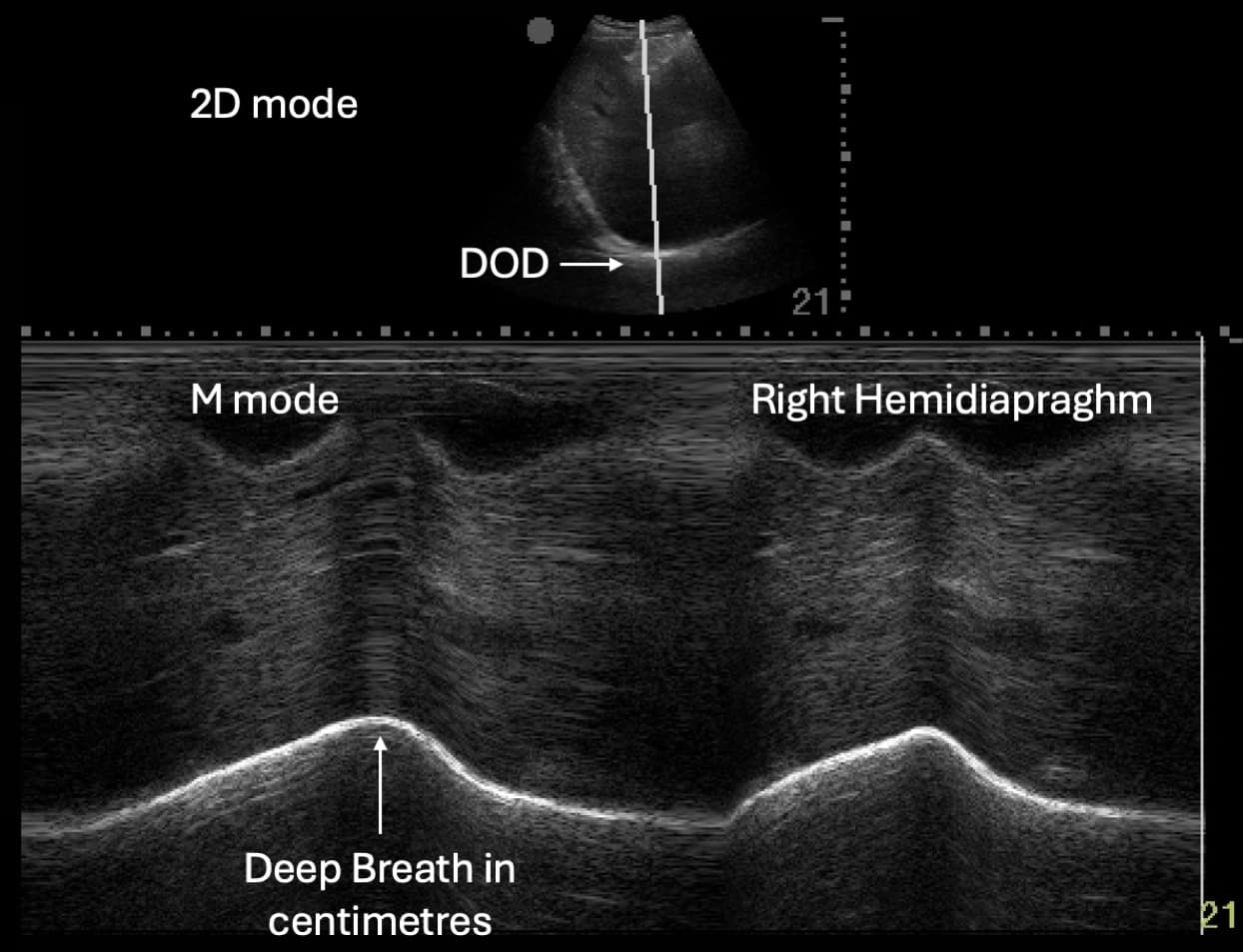
Patients with diaphragmatic paralysis will demonstrate an absence or diminished caudal movement (diaphragmatic excursion) during deep inhalation of less than <11 mm or have paradoxical motion.20 Paradoxical motion (in the cephalad direction) of the diaphragm may also be observed during the sniff test and with deep inspiration.10,21 Paradoxical motion is caused by the increasing negative pressure that is created due to the descent of the functioning diaphragm in the contralateral pleural cavity. Paradoxical movements of an unparalyzed diaphragm have also been reported in conditions such as hydrothorax, negative pressure pneumothorax, lung fibrosis, atelectasis, and subphrenic abscess.22 There are several diaphragmatic deformities or adjacent pathologies that may impact scanning of the diaphragm, including scalloping, eventration, diaphragmatic slips, inversion caused by a mass or fluid in the thorax, or peritoneal fluid.2
Zone of Apposition (ZOA) Thickening Analysis
This approach was first described by Wait et al.23 The method was further refined as the ABCDE method.24This approach is easy to learn, fast to perform and, unlike the DOD excursion analysis, provides reliable acoustic windows bilaterally, regardless of the laterality or BMI.25
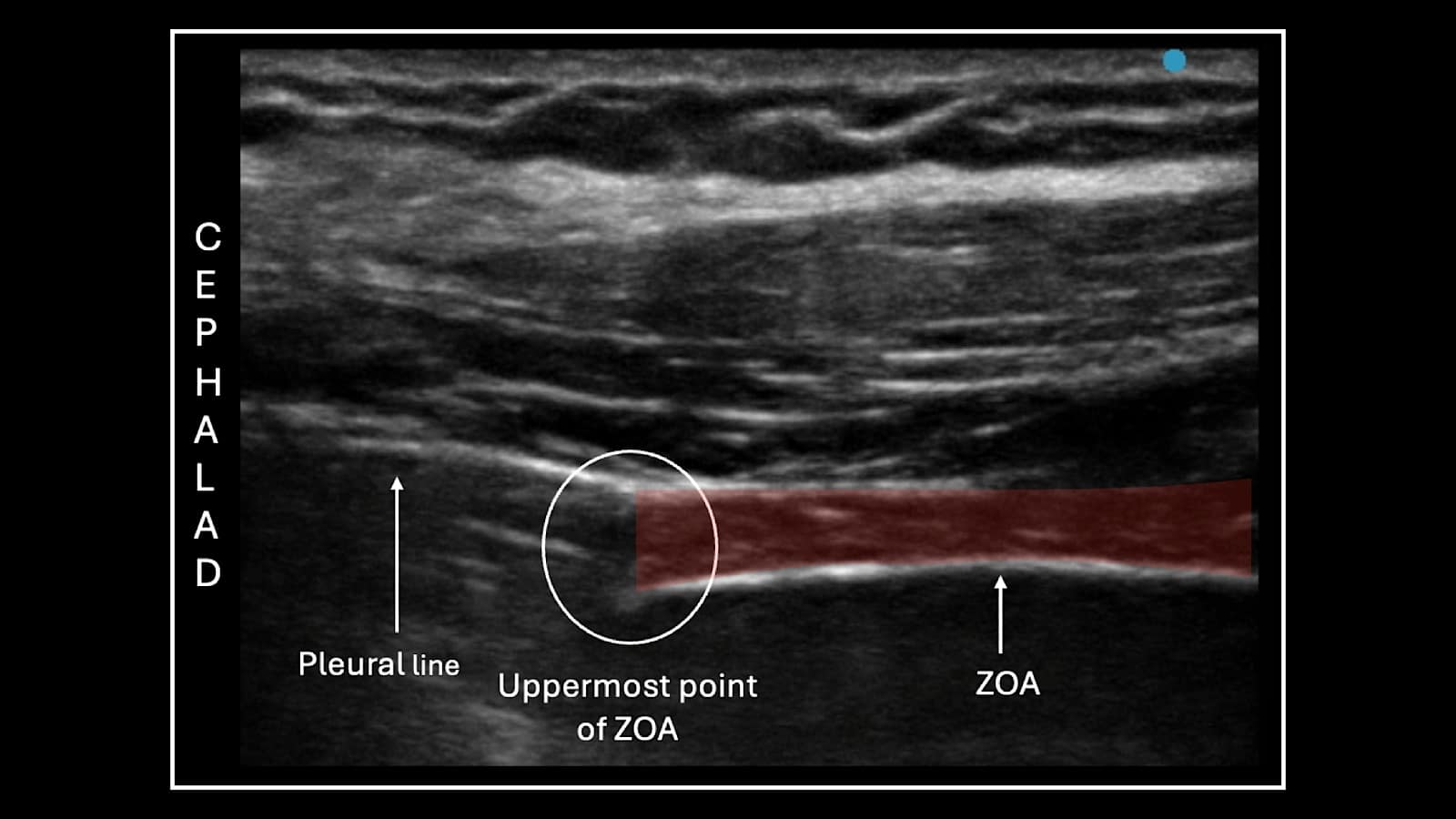
The ZOA is the area in the lower portion of the thorax where there is no lung tissue between the parietal pleura of the chest and the diaphragm is in direct apposition with the thoracic wall (Figure 5). This allows for an acoustic window with a direct view of the lateral aspect of the diaphragm. The diaphragm lies deep to the chest wall and has a characteristic three-layer pattern on ultrasound. There is a central muscular layer that is hypoechoic with a hyperechoic reticular pattern, and it is sandwiched between two hyperechoic layers, which represent the peritoneum and the parietal pleura. The area where both the lung sliding and the diaphragm can be viewed in a single image is the cephalad border or uppermost point of the ZOA (Figure 6). This allows for visualization of the cephalad aerated lung (with lung sliding), which obscures the visualization of the diaphragm and the ZOA caudal to the lung sliding where the diaphragm will be visible (transition point). This is the area where the measurements are taken.
Reproduced with permission from Giron Arango Medicine Professional Corporation.
In the ABCDE method, the patient should be placed in a semi-recumbent position. A linear ultrasound transducer (> 8 MHz) is placed between the mid-Axillary line and typically, a nerve (or equivalent) preset will be adequate. The transducer indicator is generally positioned cephalad. Visualize the Breathing (lung sliding) through the intercostal spaces at the 6th to 8th rib. Follow the lung sliding Caudally until the diaphragm is not hidden by the lung sliding at the end of a maximal inhalation and identify the Diaphragmatic thickening. During the inhalation phase of a tidal volume, the transition point of the ZOA will normally move caudally, and it is followed to its most caudal position at the end of deep exhalation. You can then perform an Examination to measure the diaphragmatic thickness and thickening fractions.
The equation of thickening fraction percent is:
/screenshot-2025-07-29-at-10-58-55-am.png?sfvrsn=6584853_1)
where Tdi is the maximum diaphragm thickness at end inhalation in the ZOA, Tde is the diaphragm thickness after passive exhalation in the ZOA, and TFd% is the diaphragm thickening fraction.
The measurement can be performed either with an M-mode scan or with two B-mode still images with calipers on each of the two images at end of deep inhalation and end exhalation, respectively (Figure 7). Most ultrasound machines will allow you to scroll through a saved loop and save still images. The transducer should be placed perpendicular to the thoracic cavity to ensure that the diaphragm borders are at their most echogenic and that the thickness is measured accurately. Rotating the transducer to be parallel with an intercostal space can limit diaphragm visualization, which may be obstructed by rib shadows.
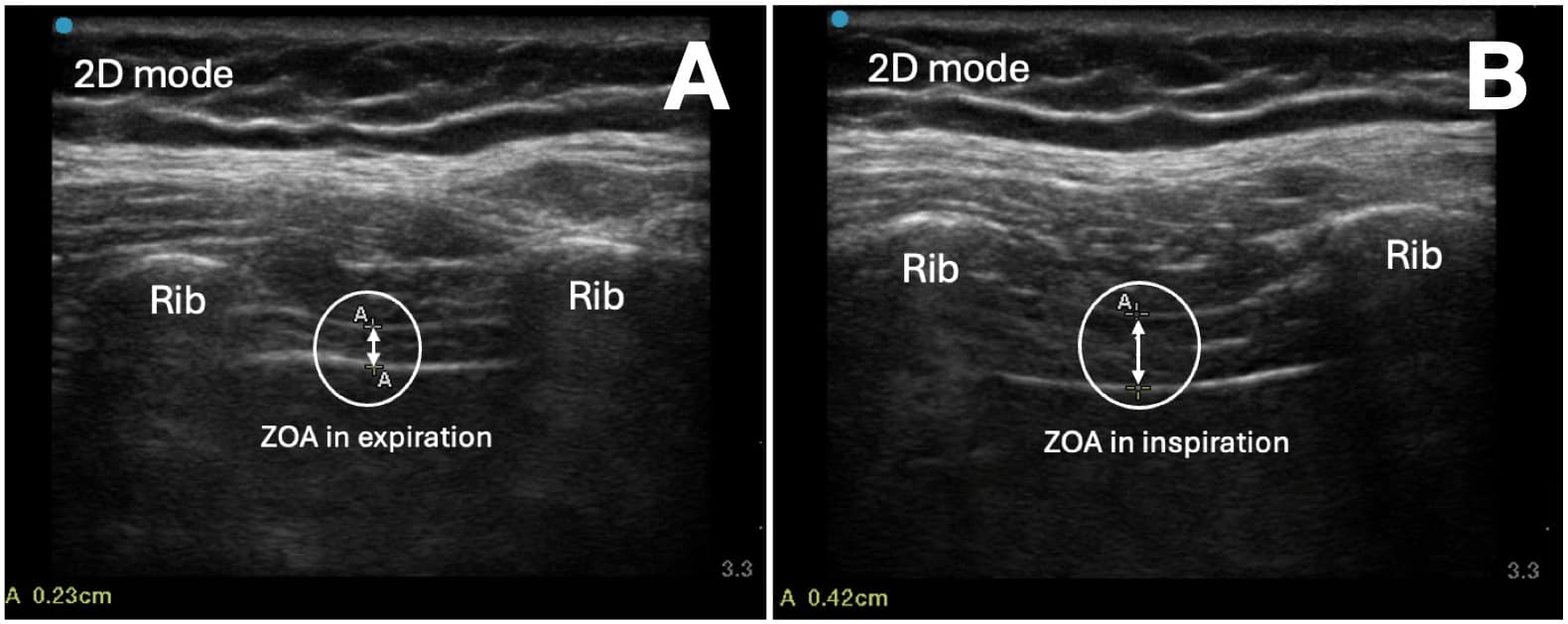
In healthy subjects, the mean TF% during quiet breathing was 50.0% (SD=25.9) and 110.7% (SD=44.3) during deep breathing.26 During a maximal inspiratory effort, TF% typically ranges between 30% and 130%.27-29 In many cases, a visual assessment can demonstrate a thickening fraction of 100%, and in such cases, formal measurement and calculation may not be required. During quiet respiration, up to one-third of patients will have little or no thickening.29
Diaphragmatic paresis is present if the TF% is < 20%,20,27,28 and diaphragm atrophy is present if the Tdi is < 0.2cm.9,27 Diaphragm thickening analysis has a high sensitivity (93%) and specificity (100%) for diaphragmatic dysfunction30 along with a high intra-rater and inter-rater reliability.28 In patients that are mechanically ventilated, diaphragm thickening is an indicator of diaphragm effort and dysfunction.31 It has been used to predict extubation success in critically ill patients.32
Zone of Apposition (ZOA) Excursion Analysis
This method has been described as a simpler and more rapid means of assessing the diaphragm,33 but others have suggested that this method is challenging to reproduce reliably.2
This method utilizes the same ZOA for evaluation that is assessed during the zone of apposition muscle thickening approach. But in this case, instead of evaluating the diaphragm muscle, the location of the uppermost point of the ZOA (transition point) is interrogated (where the aerated lung meets the ZOA). During normal inhalation, the transition point is displaced caudally. A deep inhalation accentuates this caudal movement. The relative position of the transition point in relation to the chest surface is evaluated. The caudal aspect of the visceral pleural line can normally be seen descending by about two intercostal levels during deep inhalation. In the presence of diaphragmatic paresis, there is minimal to zero movement.
This method only allows for a qualitative assessment of the diaphragm; however, a semiquantitative analysis can be performed by marking the point at the uppermost point of the ZOA on the patient’s skin during both inspiration and expiration. Two markings can be labelled on the skin with a marker at the most cephalad and most caudal position at the end of deep inhalation and at end exhalation, respectively. A tape measure can be used to find the distance between the two lines. This technique has shown to be equally successful on both sides of the thorax.16,33
Management
Unilateral diaphragmatic paresis is frequently asymptomatic in 92%-96% of healthy adults,34 and even in those with symptoms, it can frequently be managed conservatively. However, in cases where assessment, intervention, or reassessment are required, ultrasound can be a useful bedside assessment tool for the clinician. The ZOA methods assessing both excursion and thickening are a simple and reliable combination to get a rapid, reliable, and objective assessment. The ZOA thickening fraction can even be done in patients with supported modes of positive pressure ventilation. Patients with subcutaneous emphysema, chest tubes, or surgical dressings in the ZOA may not have acoustic windows in the lateral chest and may require DOD assessment. A summary of the assessment windows and clinical cut-off values is listed in Table 2.
| Imaging Area | Transducer | Measurement | Cut Off |
| Subcostal |
| Diaphragmatic Excursion |
|
| Zone of Apposition | Linear, High Frequency | Diaphragmatic Thickening Fraction | Thickening Fraction < 20% |
| Zone of Apposition | Linear, High Frequency | Diaphragmatic Excursion | Minimal excursion or paradoxical motion observed |
Conclusion
Ultrasound assessment of the diaphragm is a reproducible, reliable, and non-invasive tool that provides valuable real-time information about diaphragmatic function during perioperative care and management of mechanically ventilated patients. The combination of excursion (at the DOD and ZOA) and thickening fraction (at the ZOA) enables a comprehensive understanding of diaphragmatic performance. Whether assessing preoperative risk, troubleshooting postoperative respiratory compromise, assessing diaphragmatic function on patients on mechanical ventilation for dysfunction and titration of ventilatory support, or evaluating block-related complications, diaphragm ultrasound empowers anesthesiologists to make informed, patient-specific decisions. As its utility continues to expand beyond the critical care unit, this application of point-of-care ultrasound enhances our ability to deliver safer, more individualized perioperative care.



References
- Cohen WH. Evaluation of the diaphragm by a subcostal B-scan technique. UltrasonoGraphia Media Proceedings of the First WorId Congress on Ultrasound Diagnostics in Medicine and SIDUO III, Vienna, Austria. 1969:63.
- Sarwal A, Walker FO, Cartwright MS. Neuromuscular ultrasound for evaluation of the diaphragm. Muscle Nerve 2013;47(3):319-29. https://doi.org/10.1002/mus.23671
- Manson W HR. How I do it: lung ultrasound. ASRA Pain Medicine News 2022;47. https://doi.org/10.52211/asra020122.005
- Standring S. Respiratory diaphragm and phrenic nerves. In: Standring S, ed. Gray's Anatomy. 42nd ed. London, England: Elsevier Limited; 2021.
- Gierada DS, Curtin JJ, Erickson SJ, et al. Diaphragmatic motion: fast gradient-recalled-echo MR imaging in healthy subjects. Radiology 1995;194(3):879-84. https://doi.org/10.1148/radiology.194.3.7862995
- ATS/ERS Statement on respiratory muscle testing. Am J Respir Crit Care Med 2002;166(4):518-624. https://doi.org/10.1164/rccm.166.4.518
- Chetta A, Rehman AK, Moxham J, Carr DH, Polkey MI. Chest radiography cannot predict diaphragm function. Respir Med Jan 2005;99(1):39-44. https://doi.org/10.1016/j.rmed.2004.04.016
- Krasna MJ, Forti G. Nerve injury: injury to the recurrent laryngeal, phrenic, vagus, long thoracic, and sympathetic nerves during thoracic surgery. Thorac Surg Clin 2006;16(3):267-75, vi. https://doi.org/10.1016/j.thorsurg.2006.05.003
- Summerhill EM, El-Sameed YA, Glidden TJ, et al. Monitoring recovery from diaphragm paralysis with ultrasound. Chest 2008;133(3):737-43. https://doi.org/10.1378/chest.07-2200
- Spadaro S, Grasso S, Dres M, et al. Point of care ultrasound to identify diaphragmatic dysfunction after thoracic surgery. Anesthesiology 2019;131(2):266-78. https://doi.org/10.1097/aln.0000000000002774
- Kot P, Granell M, Rodriguez P, et al. Diaphragmatic ultrasound: early diagnosis when phrenic injury in thoracic surgery is suspected. J Cardiothorac Vasc Anesth 2018;32(6):e7-8. https://doi.org/10.1053/j.jvca.2018.06.024
- Kim DH, Lin Y, Beathe JC, et al. Superior trunk block: a phrenic-sparing alternative to the interscalene block: a randomized controlled trial. Anesthesiology 2019;131(3):521-33. https://doi.org/10.1097/aln.0000000000002841
- de la Quintana Gordon FB, Nacarino Alcorta B, Fajardo Pérez M. Ultrasound evaluation of diaphragm function and its application in critical patients, mechanical ventilation and brachial plexus block. Rev Esp Anestesiol Reanim 2017;64(9):513-21. Valoración ecográfica de la función diafragmática y sus aplicaciones en el paciente crítico, en ventilación mecánica y en la anestesia del plexo braquial. https://doi.org/10.1016/j.redar.2017.01.001
- Berg AA, Flaherty JM, Habeck JM, et al. Evaluation of diaphragmatic function after interscalene block with liposomal bupivacaine: a randomized controlled trial. Anesthesiology 2022;136(4):531-41. https://doi.org/10.1097/aln.0000000000004118
- McCool FD, Tzelepis GE. Dysfunction of the diaphragm. N Engl J Med 2012;366(10):932-42. https://doi.org/10.1056/NEJMra1007236
- Chong M, Girón-Arango L, Wild K, et al. Point-of-care ultrasound of the diaphragm after proximal brachial plexus block for shoulder surgery: a prospective observational study. Reg Anesth Pain Med2025. https://doi.org/10.1136/rapm-2024-106222
- Boussuges A, Gole Y, Blanc P. Diaphragmatic motion studied by m-mode ultrasonography: methods, reproducibility, and normal values. Chest 2009;135(2):391-400. https://doi.org/10.1378/chest.08-1541
- Bruck O, Naofal A, Senussi MH. Lung, pleura, and diaphragm point-of-care ultrasound. Semin Ultrasound CT MR 2024;45(1):120-31. https://doi.org/10.1053/j.sult.2024.01.001
- Jung KJ, Park JY, Hwang DW, et al. Ultrasonographic diaphragmatic motion analysis and its correlation with pulmonary function in hemiplegic stroke patients. Ann Rehabil Med 2014;38(1):29-37. https://doi.org/10.5535/arm.2014.38.1.29
- Sferrazza Papa GF, Pellegrino GM, Di Marco F, et al. A review of the ultrasound assessment of diaphragmatic function in clinical practice. Respiration 2016;91(5):403-11. https://doi.org/10.1159/000446518
- Lloyd T, Tang YM, Benson MD, et al. Diaphragmatic paralysis: the use of M mode ultrasound for diagnosis in adults. Spinal Cord 2006;44(8):505-8. https://doi.org/10.1038/sj.sc.3101889
- Alexander C. Diaphragm movements and the diagnosis of diaphragmatic paralysis. Clin Radiol1966;17(1):79-83. https://doi.org/10.1016/s0009-9260(66)80128-9
- Wait JL, Nahormek PA, Yost WT, et al. Diaphragmatic thickness-lung volume relationship in vivo. J Appl Physiol (1985) 1989;67(4):1560-8. https://doi.org/10.1152/jappl.1989.67.4.1560
- Tsui JJ, Tsui BC. A novel systematic ABC approach to diaphragmatic evaluation (ABCDE). Can J Anaesth 2016;63(5):636-7. https://doi.org/doi:10.1007/s12630-015-0566-x
- Khurana J, Gartner SC, Naik L, et al. Ultrasound identification of diaphragm by novices using ABCDE technique. Reg Anesth Pain Med 2018;43(2):161-5. https://doi.org/10.1097/aap.0000000000000718
- Yamada T, Minami T, Yoshino S, et al. Relationship between diaphragm thickness, thickening fraction, dome excursion, and respiratory pressures in healthy subjects: an ultrasound study. Lung2024;202(2):171-8. https://doi.org/10.1007/s00408-024-00686-2
- Gottesman E, McCool FD. Ultrasound evaluation of the paralyzed diaphragm. Am J Respir Crit Care Med 1997;155(5):1570-4. https://doi.org/10.1164/ajrccm.155.5.9154859
- Boon AJ, Harper CJ, Ghahfarokhi LS, Strommen JA, Watson JC, Sorenson EJ. Two-dimensional ultrasound imaging of the diaphragm: quantitative values in normal subjects. Muscle Nerve2013;47(6):884-9. https://doi.org/10.1002/mus.23702
- Harper CJ, Shahgholi L, Cieslak K, et al. Variability in diaphragm motion during normal breathing, assessed with B-mode ultrasound. J Orthop Sports Phys Ther 2013;43(12):927-31. https://doi.org/10.2519/jospt.2013.4931
- Boon AJ, Sekiguchi H, Harper CJ, et al. Sensitivity and specificity of diagnostic ultrasound in the diagnosis of phrenic neuropathy. Neurology 2014;83(14):1264-70. https://doi.org/10.1212/wnl.0000000000000841
- Umbrello M, Formenti P, Longhi D, et al. Diaphragm ultrasound as indicator of respiratory effort in critically ill patients undergoing assisted mechanical ventilation: a pilot clinical study. Crit Care2015;19(1):161. https://doi.org/10.1186/s13054-015-0894-9
- Blumhof S, Wheeler D, Thomas K, et al. Change in diaphragmatic thickness during the respiratory cycle predicts extubation success at various levels of pressure support ventilation. Lung2016;194(4):519-25. https://doi.org/10.1007/s00408-016-9911-2
- Da Conceicao D, Perlas A, Giron Arango L, et al. Validation of a novel point-of-care ultrasound method to assess diaphragmatic excursion. Reg Anesth Pain Med 2024;49(11):800-4. https://doi.org/10.1136/rapm-2023-104983
- Kim KS, Ahn JH, Yoon JH, et al. Hemidiaphragmatic paresis following interscalene brachial plexus block with 2-point injection technique. Pain Physician 2021;24(8):507-15.