How I Do It: Denervation of the Hip Joint Under Ultrasound and Fluoroscopy
Cite as: Almazidi A, Machi A, Meng H. How I do it: Denervation of the hip joint under ultrasound and fluoroscopy. ASRA Pain Medicine News 2025;50. https://doi.org/10.52211/asra080125.007.
How I Do It
Introduction
There is a 7%-10% prevalence of hip pain in adults 45 years or older, with the leading cause of chronic hip pain being osteoarthritis.1 Other causes of chronic hip pain include rheumatoid arthritis, labral tears, osteonecrosis of the femoral head, avascular necrosis, post-traumatic arthritis, acetabular fractures, chronic infections, and persistent pain after total hip replacement. Irrespective of the pathology, hip pain negatively impacts mobility and overall quality of life. Hip arthroplasty is one of the most common and successful types of orthopedic surgeries. However, concerns regarding patient comorbidities and perioperative risk may deter some patients from hip replacement surgery or make them poor surgical candidates.2 The life-span of the implanted hardware is also limited to between 15-25 years, which results in patients typically being deferred for hip arthroplasty until they are well into their 50s or 60s.3 Consequently, there is a need for safe and effective procedural intervention for the management of hip joint pain in these patients.
Innervation of the Hip Joint Capsule
The majority of pain in the hip joint originates from the joint capsule, which is richly innervated by the articular nerves branching from multiple nerves. The anterior hip capsule, which is the target of discussion, is innervated by articular branches of the femoral, obturator, and accessory obturator nerves, which are well described by Short et al.4 The posterior hip capsule is innervated by the quadratus femoris, superior gluteal, and perhaps the sciatic and inferior gluteal nerves.5
Previously, the femoral nerve was thought to innervate the superolateral and inferolateral aspects of the joint capsule, whereas the superomedial and inferomedial aspects of the joint capsule were innervated by the obturator nerve and the accessory obturator nerves.6 The systematic review and meta-analysis by Tomlinson et al. offers an updated summary pictograph of the innervation discussed (see Figure 1).7
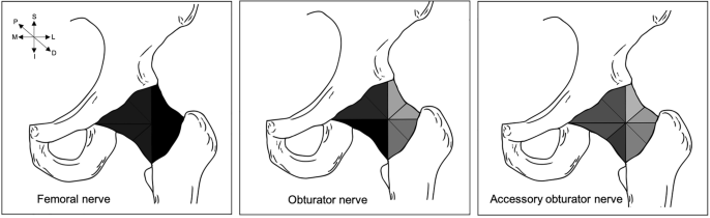
Reproduced with open access from Tomlinson et al. Sci Rep 2021.
Indications and Patient Selection
Hip denervation is indicated in patients with chronic hip pain (pain for more than 3 months), who have failed to respond to conservative management and are not surgical candidates or do not want to pursue surgery.8 Any pathology with the potential to cause pain in the hip joint, which may include, but is not limited to osteoarthritis, rheumatoid arthritis, avascular necrosis, persistent pain after total hip replacement, post-traumatic arthritis, labral tears of acetabulum, and acetabular fractures, may be an underlying cause of the chronic hip pain. In the authors’ practices, certain subgroups of patients present most commonly: those who have experienced lower extremity trauma (ie, femur fracture, acetabular fracture), those with advanced osteoarthritis who are too frail or ill to undergo hip arthroplasty, and those who have had hip arthroplasty but continue to have persistent pain. Other patient groups that frequently benefit from hip denervation include patients with sickle cell disease or significant steroid use resulting in avascular necrosis of the hip. No specific exclusion criteria exist outside of the typical contraindications (ie localized infection, severe coagulopathy, etc.) with respect to hip denervation.
Ultrasound-Guided Hip Denervation
Hip denervation was originally described as a technique under fluoroscopic guidance. A previous HIDI article on fluoroscopic-guided hip denervation can be found here.
The targets of the articular branches of the femoral nerve, accessory obturator nerve, and obturator nerve are well established on ultrasound.4 Sonographically, the reliably targeted articular branches of the femoral nerve traverse between the inferior and medial portion of the anterior inferior iliac spine (AIIS) and along the iliopubic eminence (IPE). A suggestion for the radiofrequency (RF) cannula entry point being 1-2 cm lateral to the femoral artery has been made in the literature; however, it has been the authors’ experience to insert the RF cannula just medial to the AIIS and along the same angle as the bony descent from the AIIS to the IPE.6 This approach allows for improved angulation to place the needle deep to the psoas tendon without piercing the structure.
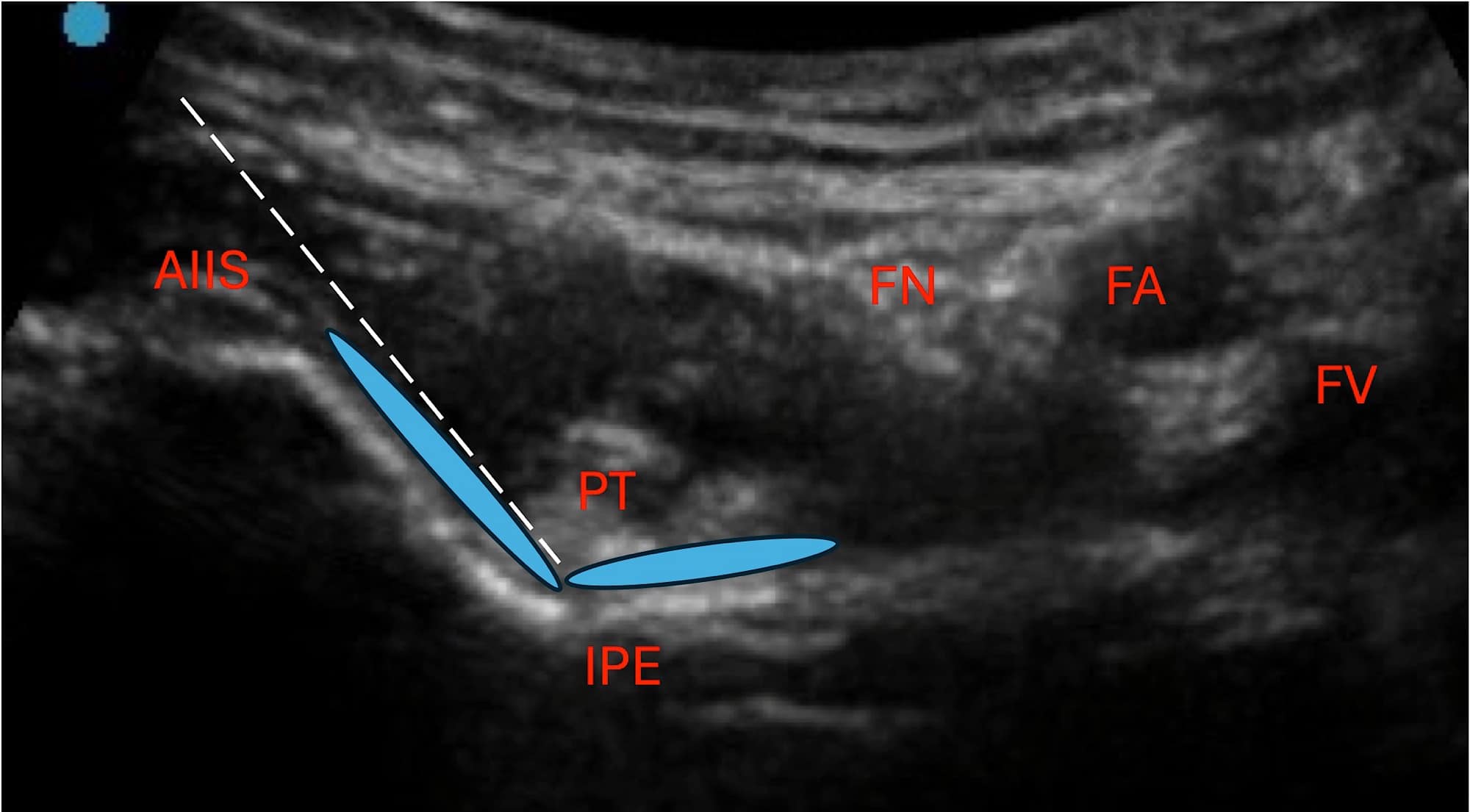
AIIS = anterior inferior iliac spine, PT = psoas tendon, IPE = iliopubic eminence, FN = femoral nerve, FA = femoral artery, FV = femoral vein
Sequential lesion along the target zone can also be performed easily by withdrawing the RF cannula more proximally. The articular branches of the accessory obturator nerve are also best targeted along the IPE and are typically captured via the lesioning approach described for the articular branches of the femoral nerve. The target for the articular branches of the obturator nerve can be visualized along the inferomedial acetabulum (IMA). This view is typically first achieved through the hip intra-articular view sonographically. The probe is then translated inferomedially to visualize the inferomedial acetabulum.
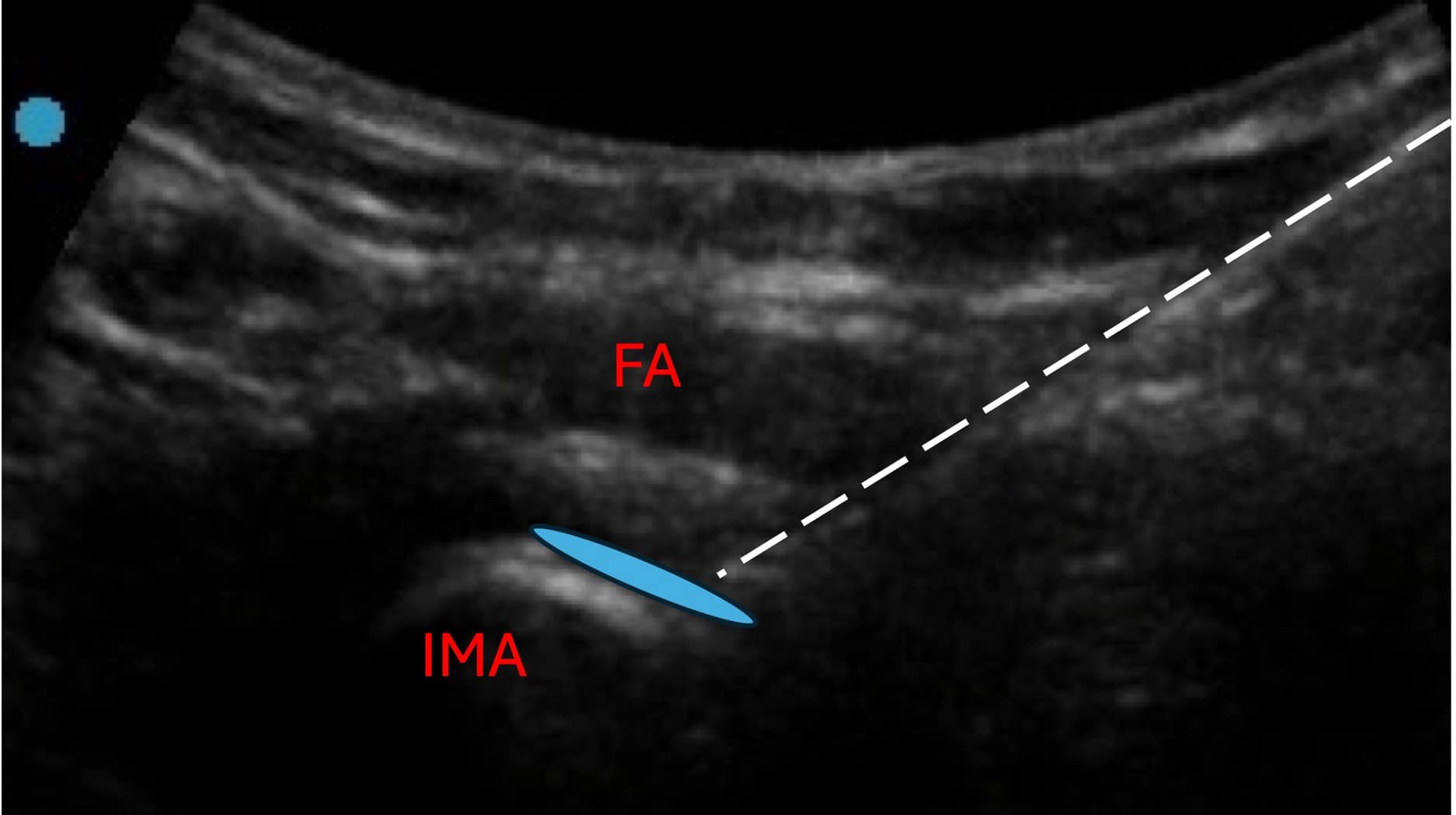
IPE = iliopublic eminence, FA = femoral artery
Benefits of ultrasound use in hip denervation include improved visualization of the vasculature, neural, and important musculoskeletal structures when traversing the needle towards the target zone. In targeting the IPE where the articular branch of the femoral nerve and the accessory obturator nerves lie, care should be taken to avoid RF cannula puncture of the psoas tendon, which otherwise may result in injury of the tendon and significant post-procedure pain (see Figure 2).6 In targeting the inferomedial acetabulum where the articular branches of the obturator nerve lie, care should be taken to avoid RF cannula through the femoral artery and femoral nerve as well as adjacent vasculature, such as the external pudendal artery.6 Although the rates of femoral artery and nerve injuries are not well known for hip denervation, a sonographic approach serves to reduce the risks of inadvertent injury (see Figure 3).
A strict sonographic-only approach to hip denervation allows for a relatively low cost of imaging modality compared to fluoroscopy. In combination with chemical ablation using neurolytics of these target nerves, hip denervation can be performed to good effect with very modest equipment cost.2
Combined Ultrasound and Fluoroscopic Hip Approach
The goal of a combined ultrasound fluoroscopic approach is to improve accuracy, efficiency, and safety in performing hip denervation. It is the preferred method of the authors. The typical setup is to have the fluoroscope come across the patient and with the ultrasound machine placed along the foot of the procedure table.
For the articular branches of the femoral nerve and accessory obturator nerve, the RF cannula is inserted under ultrasound guidance from lateral to medial with modest cephalad to caudad angulation from the AIIS to the IPE at an overall angulation corresponding to the bony descent. The RF cannula is placed with the tip deep to the psoas tendon or medial, bypassing the psoas tendon with continuous ultrasound guidance. A larger lesion is preferred to ensure capture of the target nerves due to the variable innervation patterns of the anterior capsule.9 The author HM typically uses a bipolar approach, and therefore, a second RF cannula is inserted just cephalad to the first cannula along the IPE. The author AM uses a cooled radiofrequency ablation technique.
Once the RF cannulas have been brought into contact with IPE, fine-tuning the cannula's position under fluoroscopic guidance is performed to ensure optimal placement of RF cannulas to one another (see Figures 4A and 4B).
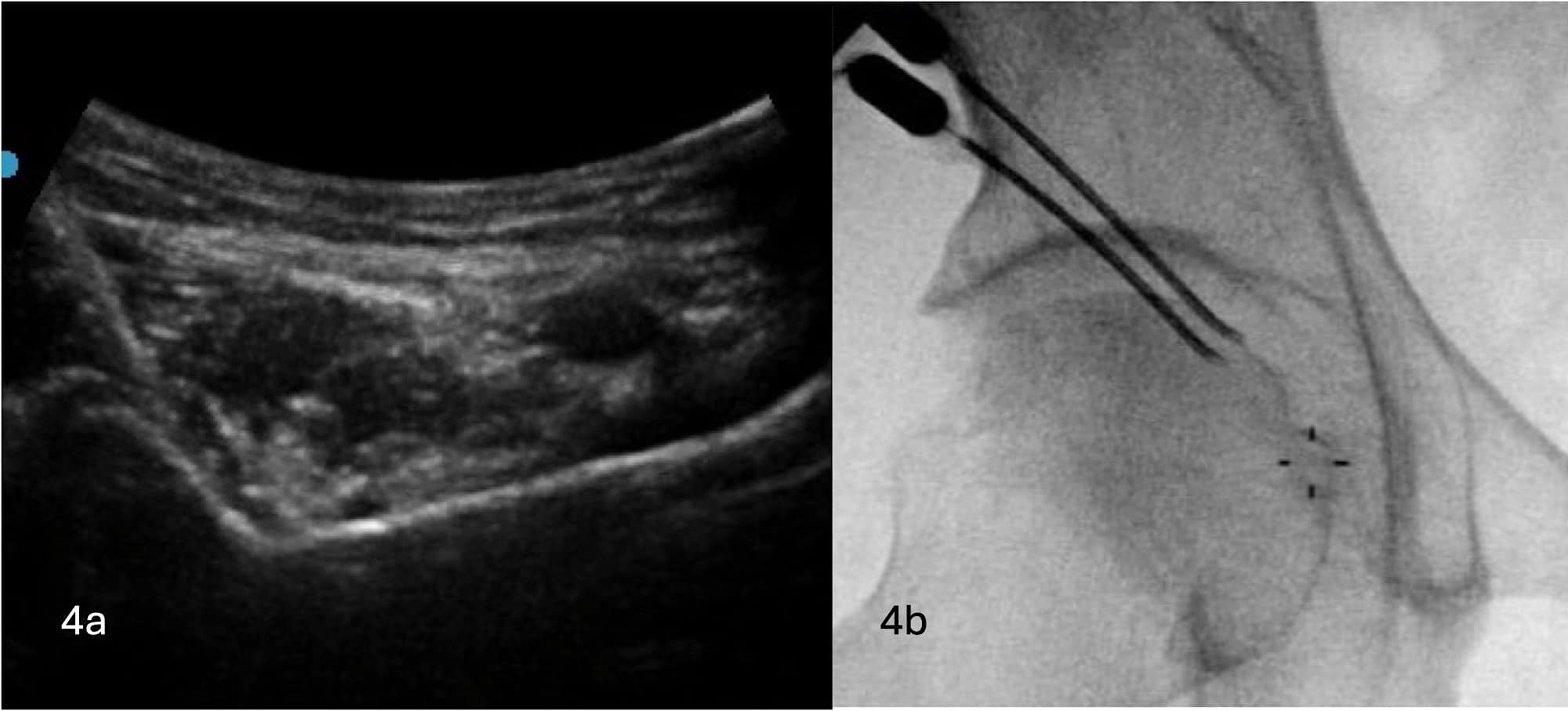
Sequential bipolar RF is carried out with the cannulas withdrawn slightly each time to ensure the entirety of the area between AIIS and IPE is covered (see Figures 5A and 5B).
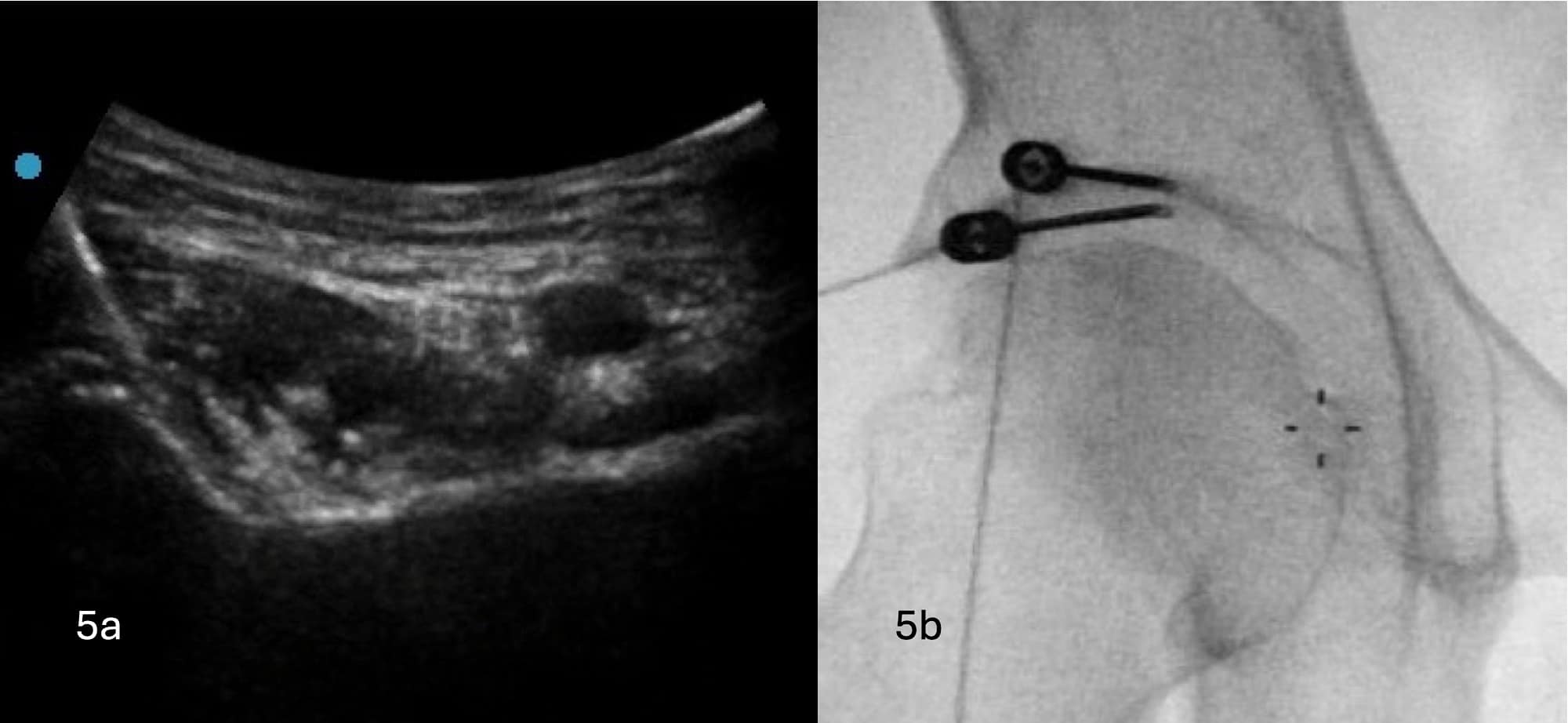
Figure 5A. Ultrasound image demonstrating withdrawn RF cannula compared to Figure 4. Figure 5B. Fluoroscopic image demonstrating the corresponding position of the withdrawn RF cannula.
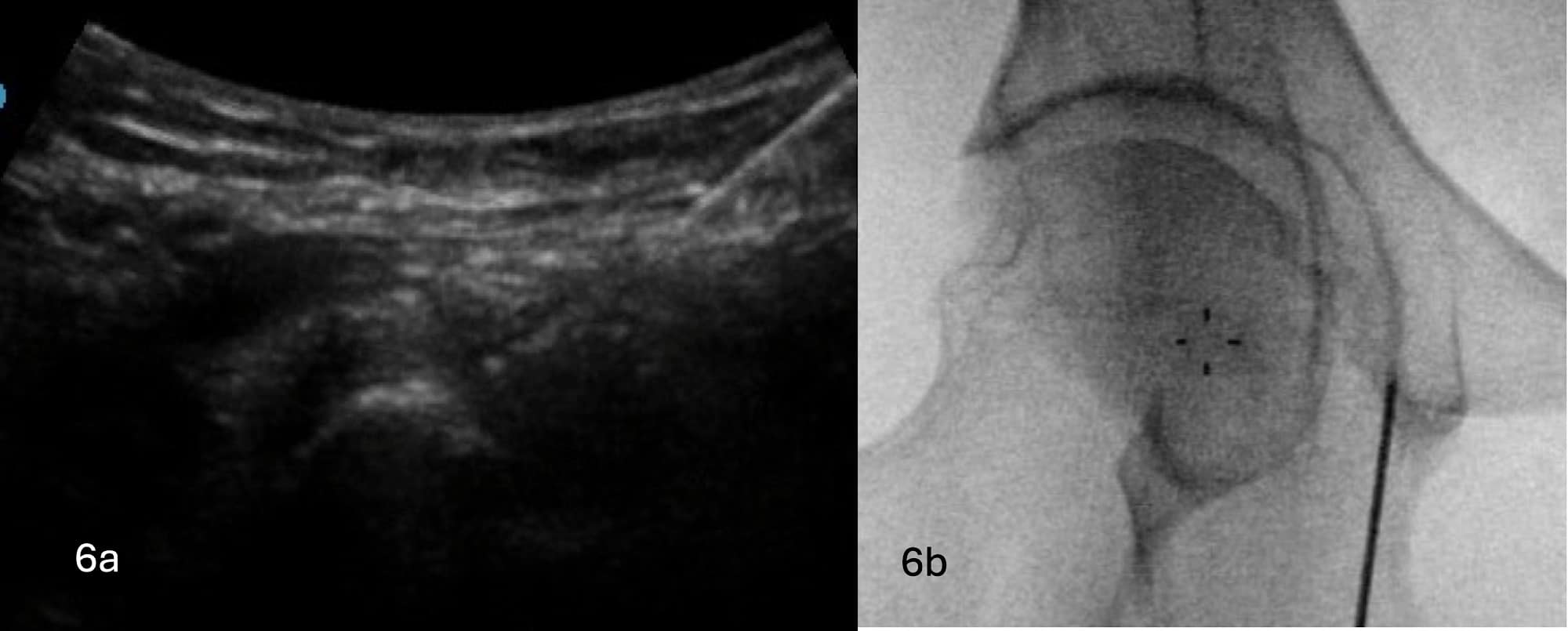
For the articular branches of the obturator nerve, author HM inserts the RF cannula sonographically from lateral to medial, caudad to cephalad, towards the IMA. The author positions the patient with slight hip external rotation and inserts the RF cannula in a more longitudinal fashion medially, through the adductor muscles, to avoid the femoral and lateral circumflex femoral arteries. Upon contact with bone, fluoroscopy is then used to ensure optimal placement of the RF cannula (see Figures 6A and 6B). Depending on the angle of insertion towards the IMA, the RF cannula may contact the bone further along the pubis and will need to be optimized. The author uses monopolar RF with two to three lesions with the RF cannula in slightly different angles on the IMA to ensure larger area of lesioning whereas the author uses cooled radiofrequency ablation.
Conclusion
Both fluoroscopic and ultrasound-guided hip denervation techniques have been described. The authors’ opinions are that a combined ultrasound and fluoroscopy-guided technique allows for improved accuracy, efficiency, and safety in performing hip denervation. For further reading, there have been several articles published on the use of dual-modality imaging guided techniques including Kapural et al.,10 Kumar et al.,6 and a recently published review article by Pun et al.8
Acknowledgement
We would like to acknowledge the open access use of Figure 1 from Tomlinson J, Ondruschka B, Prietzel T, et al. A systematic review and meta-analysis of the hip capsule innervation and its clinical implications. Sci Rep 2021;11(1):5299. https://doi.org/10.1038/s41598-021-84345-z.



References
- Murphy L, Helmick CG. The impact of osteoarthritis in the United States: a population-health perspective: a population-based review of the fourth most common cause of hospitalization in U.S. adults. Orthop Nurs 2012;31(2):85-91. https://doi.org/10.1097/NOR.0b013e31824fcd42
- Kwun-Tung Ng T, Chan WS, Peng PWH, et al. Chemical hip denervation for inoperable hip fracture. Anesth Analg 2020;130(2):498-504. https://doi.org/10.1213/ANE.0000000000004172
- Evans JT, Evans JP, Walker RW, et al. How long does a hip replacement last? a systematic review and meta-analysis of case series and national registry reports with more than 15 years of follow-up. Lancet2019;393(10172):647-54. https://doi.org/10.1016/s0140-6736(18)31665-9
- Short AJ, Barnett JJG, Gofeld M, et al. Anatomic study of innervation of the anterior hip capsule: implication for image-guided intervention. Reg Anesth Pain Med 2018;43(2):186-92. https://doi.org/10.1097/AAP.0000000000000701
- Nagpal AS, Brennick C, Occhialini AP, et al. Innervation of the posterior hip capsule: a cadaveric study. Pain Med 2021;22(5):1072-9. https://doi.org/10.1093/pm/pnab057
- Kumar P, Hoydonckx Y, Bhatia A. A review of current denervation techniques for chronic hip pain: anatomical and technical considerations. Curr Pain Headache Rep 2019;23(6):38. https://doi.org/10.1007/s11916-019-0775-z
- Tomlinson J, Ondruschka B, Prietzel T, et al. A systematic review and meta-analysis of the hip capsule innervation and its clinical implications. Sci Rep 2021;11(1):5299. https://doi.org/10.1038/s41598-021-84345-z
- Pun M, Ng T, Vermeylen K, et al. Innervation of the hip joint: implications for regional anaesthesia and image-guided interventional pain procedures. BJA Educ 2024;24(6):191-202. https://doi.org/10.1016/j.bjae.2024.02.005
- Bhatia A, Hoydonckx Y, Peng P, et al. Radiofrequency procedures to relieve chronic hip pain: an evidence-based narrative review. Reg Anesth Pain Med 2018;43(1):72-83. https://doi.org/10.1097/AAP.0000000000000694
- Kapural L, Jolly S, Mantoan J, et al. Cooled radiofrequency neurotomy of the articular sensory branches of the obturator and femoral nerves - combined approach using fluoroscopy and ultrasound guidance: technical report, and observational study on safety and efficacy. Pain Physician2018;21(3):279-84.