POCUS Spotlight: Transcranial Doppler
Cite as: Sai GM, Kumar S, Chhabra S, et al. POCUS spotlight: transcranial Doppler. ASRA Pain Medicine News 2025;50. https://doi.org/10.52211/asra050125.010
POCUS Spotlight
Introduction
In critically ill patients with acute brain injury, multimodal monitoring is used to diagnose and monitor various neurological complications. Transcranial Doppler (TCD) is one such tool for rapid bedside assessment and monitoring of cerebrovascular hemodynamics. Introduced by Rune Aaslid in 1982, TCD has been pivotal in the management of stroke, subarachnoid hemorrhage (SAH), and intracranial hypertension.1
Transcranial Doppler
Basic Concepts
The TCD works on the principle of the Doppler effect. A low-frequency 2 MHz transducer with a circular footprint is used for this purpose (Figure 1). The transducer is placed on thin parts of the skull called “acoustic windows” or “insonation windows” to interrogate cerebral blood vessels. A pulse wave Doppler (PWD) is used to obtain spectral waveforms of insonated blood vessels. Parameters, such as peak systolic velocity (PSV), end-diastolic velocity (EDV), mean flow velocity (MFV), and pulsatility index (PI), can be derived from the spectral waveform.
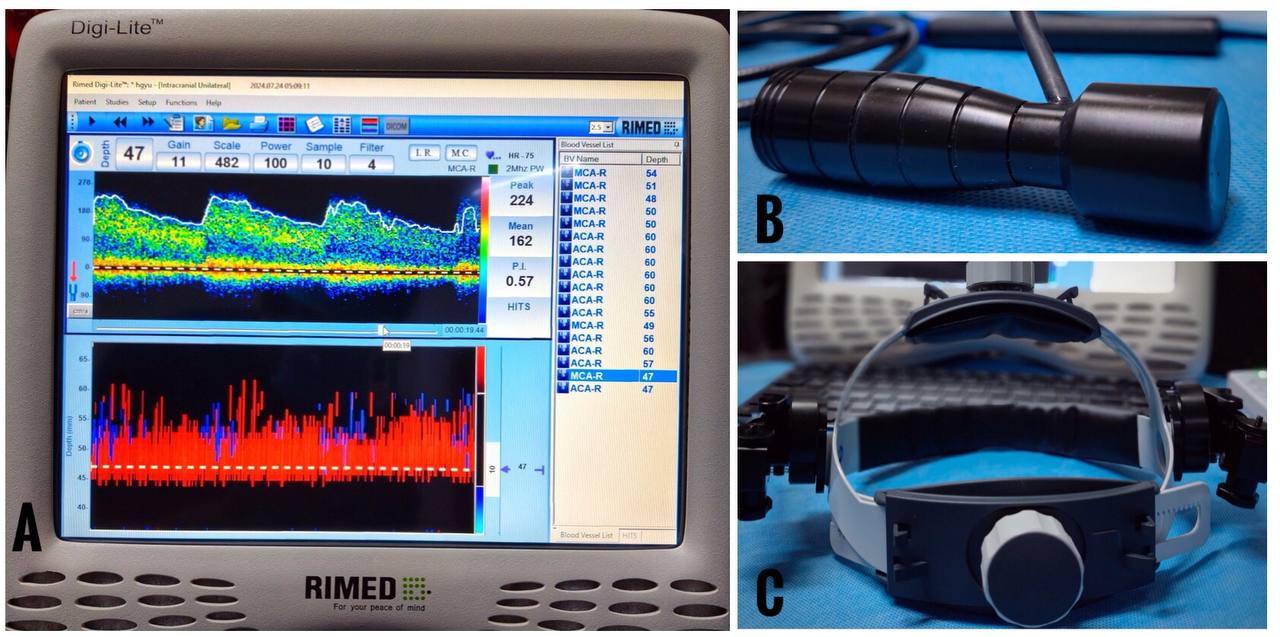
A = monitor, B =low-frequency probe with circular footprint, C = headset (manual).
Advancements in Technology
In the last three decades, TCD technology has advanced, including transcranial power motion mode (PMD), transcranial color-coded duplex sonography (TCCS), and contrast-enhanced TCCS.
Mark Moehring and Merril Spencer were the pioneers behind the innovation of the PMD. It uses multiple sample gates to display information regarding different flow signal intensities and directions. Advantages include easy identification of insonation windows, pickup of flow signals for multiple blood vessels at a stretch, and better delineation of microemboli.2
TCCS is performed using a phased-array probe. This imaging modality's advantages include visualization of brain parenchyma with B-mode, better localization of the blood vessels with color Doppler, and angle correction for PWD. One important limitation is the difficulty performing continuous monitoring with the phased-array probe. Table 1 summarizes significant differences between conventional TCD and TCCS.
| TCD | TCCS | |
| Machine | A dedicated TCD machine and specific equipment are required. | TCCS is performed with ultrasound machines available in ICUs. |
| Transducer | A low-frequency probe with a circular footprint is used. | A phased array probe is used. |
| Angle Correction | Cannot be done | Can be done |
| Color Doppler | Only available in the newer TCD machines | Blood vessels can be localized and traced with color Doppler. |
| Two-dimensional imaging | Not possible | Brain parenchyma and its pathologies can be visualized. |
| Continuous monitoring | Possible with equipment such as a headset (manual and robotic) | Not possible |
Contrast-enhanced TCCS is a method of performing TCCS after administration of intravenous ultrasound contrast agents like microbubble suspension of sulfur hexafluoride. It helps better localize blood vessels in case of poor insonation windows. Increased flow velocities are observed in normal patients after contrast administration as the contrast microbubbles are smaller than red blood cells. This might limit the interpretation of flow velocities in patients with SAH.
Cerebral Circulation
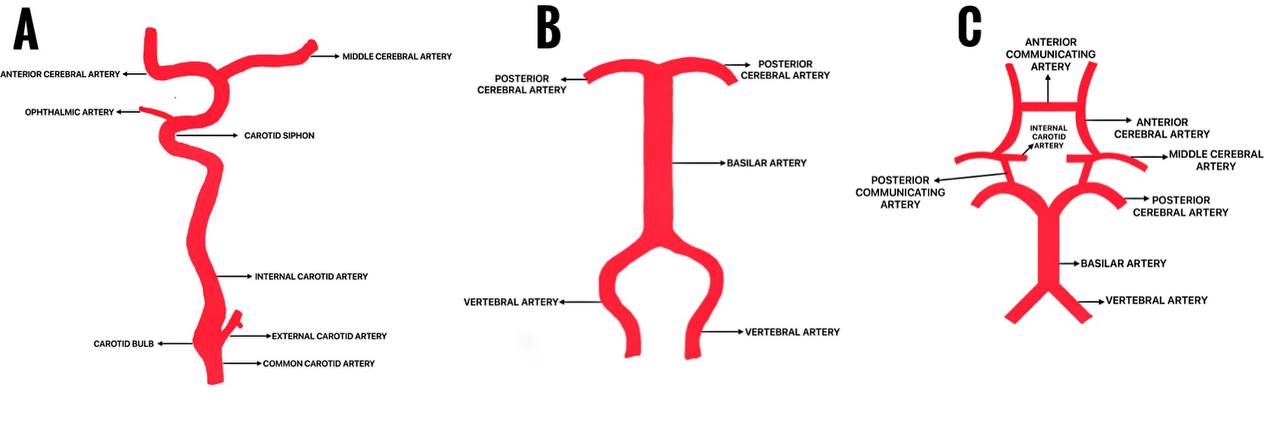
The bilateral internal carotid artery (ICA) and vertebral artery (VA) supply blood to the brain. The ICA originates from the common carotid artery (CCA) at the level of the superior border of the thyroid cartilage. Before bifurcation into the middle cerebral artery (MCA) and anterior cerebral artery (ACA) at the anterior clinoid process in the cranium, ICA navigates through the neck in the carotid sheath (cervical segment), the petrous portion of the temporal bone (petrous segment), and the cavernous sinus (cavernous segment). The ophthalmic artery (OA) arises from the ICA distal to the cavernous sinus. A portion of ICA from the beginning of the cavernous segment until the end of ICA bifurcation is S-shaped and called a carotid siphon (CS) (Figure 2A).
After originating from the subclavian artery, the vertebral artery travels through the cervical vertebrae, enters the cranium, and joins the opposite vertebral artery to form the basilar artery (BA) at the lower end of the pontomedullary junction. The basilar artery divides into the right and left posterior cerebral arteries at the midbrain (Figure 2B). The Circle of Willis at the base of the brain consists of arterial anastomosis contributed by anterior cerebral arteries, anterior communicating artery (ACOM), posterior communicating arteries (PCOM), the terminal portions of the ICA, and the posterior cerebral arteries (PCA) (Figure 2C). Such arterial anastomoses are a source of collaterals between the right and left cerebral hemispheres through the ACOM artery and the anterior and posterior circulation through the PCOM artery.
Insonation Windows
A low-frequency (2 MHz) transducer with a circular footprint insonates cerebral vessels at insonation windows. Transtemporal, suboccipital, submandibular, and transorbital windows are the four standard insonation windows utilized during TCD examination (Figure 3). Each insonation window is unique, allowing us to insonate and examine specific cerebral blood vessels (Figures 4-7). Techniques to insonate and identify cerebral blood vessels through the four standard insonation windows are summarized in Table 2.3
| Insonation window | Position of patient and transducer | Artery insonated | Depth of insonation (in mm) | Flow in relation to the transducer | Mean flow velocity (cm/sec) |
| Transtemporal | The patient is supine with the head in a neutral position. The transducer is placed on the temporal aspect of the skull, cephalad to the zygomatic arch and anterior to the tragus. The probe's anterior angulation will help in better localizing ACA. | MCA | 30-65 | Towards | 55 ± 13 |
| ACA | 60-75 | Away | 50 ± 11 | ||
| Slight inferior angulation of the transducer from the position as mentioned above. | ICA Bifurcation | 51-65 | Bidirectional | 39 ± 9 | |
| Slight posterior angulation of the transducer from the position as mentioned above for MCA. | P1 segment of PCA | 55-75 | Towards | 39 ± 10 | |
| P2 segment of PCA | 55-75 | Away | 40 ± 10 | ||
| Transorbital | The patient is supine with the head in a neutral position. The transducer is placed on the orbit and angulated in inferior and medial directions. | OA | 40-50 | Towards | 21 ± 5 |
| Same as mentioned above for OA. | CS | 58-65 | Variable | 41 ± 11 | |
| Suboccipital | The patient is in a lateral decubitus position with the head slightly flexed. The transducer is placed on the nuchal crest slightly off the midline and angulated in superior and medial directions. | VA | 40-75 | Away | 37 ± 10 |
| Slight superior angulation of the transducer from the position as mentioned above. | BA | 80-105 | Away | 41 ± 10 | |
| Submandibular | The patient is supine with the head in slight extension. The transducer is placed below the angle of the mandible and angulated in superior and medial directions. | Extracranial ICA | 35-80 | Away | 34 ± 9 |
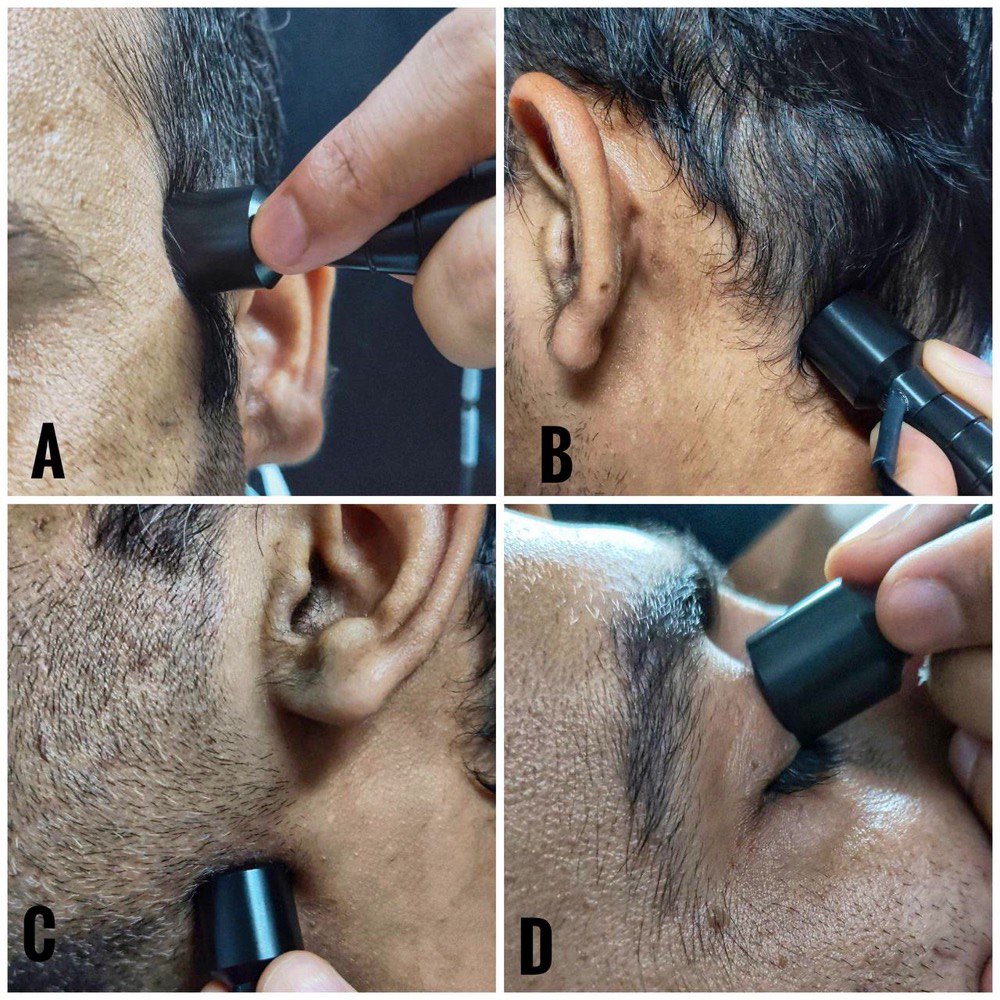
A: transtemporal window, B = suboccipital window, C - submandibular window, D = transorbital window
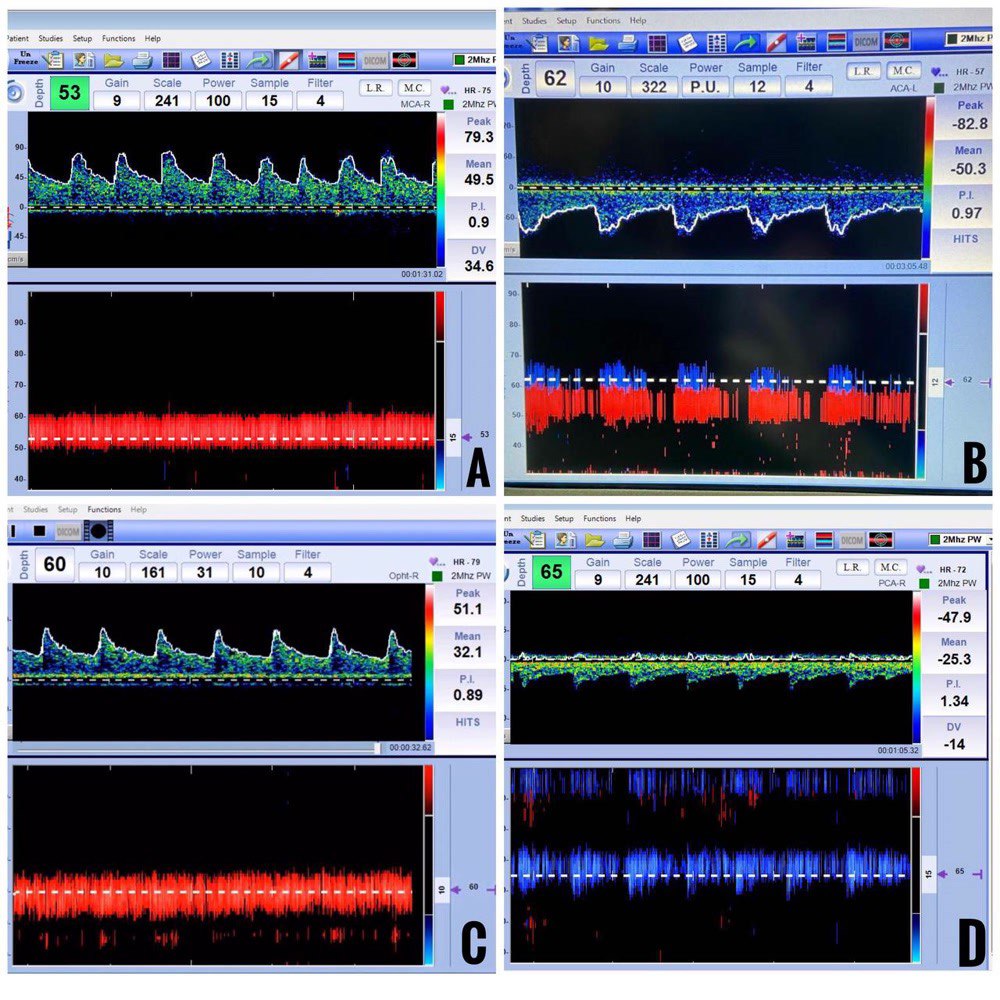
A = middle cerebral artery, B = anterior cerebral artery, C = posterior cerebral artery (P1 segment), and D = posterior cerebral artery (P2 segment).
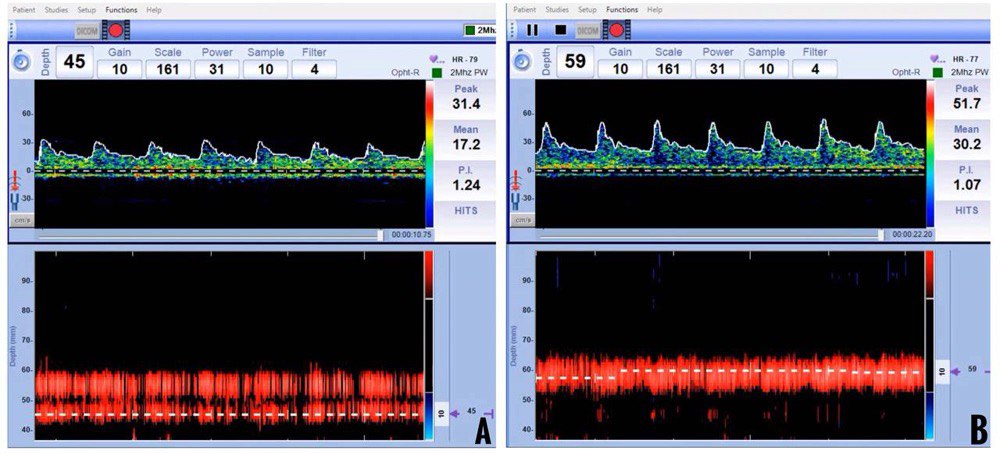
A = ophthalmic artery, B = carotid siphon
A = vertebral artery, B = basilar artery
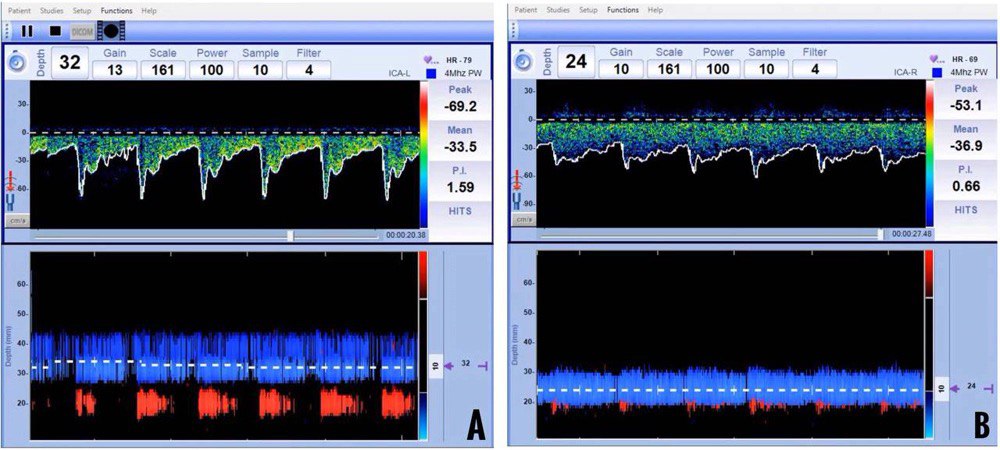
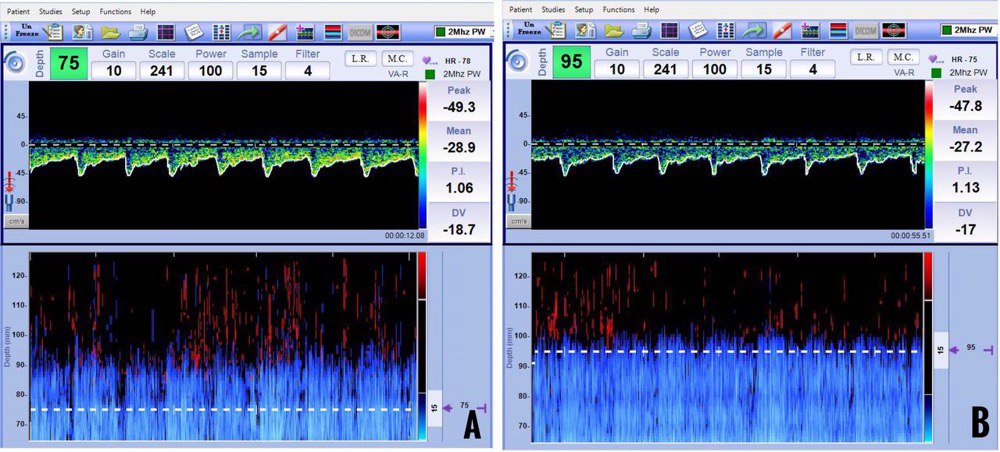
Figure 7: Spectral waveforms of arteries insonated at the submandibular window.
A = external carotid artery, B = internal carotid artery
Transcranial Doppler Protocol
In 2007, the American Society of Neuroimaging recommended a protocol for performing conventional TCD systematically. A simplified version of these recommendations has been presented as a flowchart in Figure 8.4
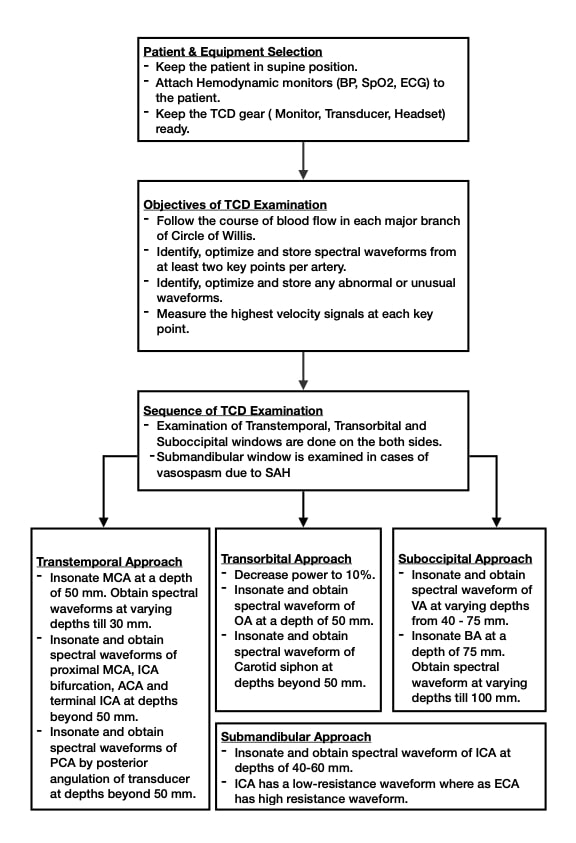
ACA = anterior cerebral artery, BA = basilar artery, ECA = external carotid artery, ICA = internal carotid artery, MCA = middle cerebral artery, OA = ophthalmic artery, PCA = posterior cerebral artery, VA = vertebral artery
Spectral Waveform Analysis
Intracranial blood flow exhibits characteristics of low-resistance circulation. It accelerates in the systole and slows down in the diastole without reaching baseline. The spectral waveform of an insonated intracranial artery shows rapid systolic upstroke, dicrotic notch, and gradual deceleration with forward flow present throughout the diastole (Figure 9). Analysis of the waveform morphology gives a rough estimate of pathologies, such as stenosis, vasospasm, and raised intracranial pressure (ICP). However, PSV, EDV, and MFV obtained from the spectral waveform provide a deeper insight into the pathophysiology. With the assumption of the cross-sectional area of the blood vessel and insonation angle being constant, the MFV is often taken as a surrogate for cerebral blood flow (CBF). MFV is more significant than systolic and diastolic velocities as it is less dependent on heart rate, contractility, and peripheral vascular resistance. However, MFV depends on various other factors influencing CBF, such as age, sex, hematocrit, partial pressure of carbon dioxide (PaCO2), collateral flows, and cerebrovascular resistance (Table 3 and Table 4).5,6 Additional parameters, such as the pulsatility index (PI), resistive index (RI), and Lindegaard ratio (LR), can be calculated by integrating velocities from the spectral waveform.7

PSV = peak systolic velocity, EDV = end diastolic velocity, S = systole, D = diastole
| Factors | Change in Mean Flow Velocity |
| Age | The decrease in velocities is significant after the fifth decade. |
| Sex | Slightly higher velocities in females |
| Pregnancy | Velocities decrease in the third trimester. |
| Partial pressure of Carbon dioxide (PaCO2) | Velocities increase with an increase in PaCO2. |
| Hematocrit | Velocities increase with a decrease in hematocrit. |
| Factors Increasing Mean Flow Velocity | Factors Decreasing Mean Flow Velocity |
| Hyperemia | Hypoperfusion |
| Vasospasm | Vessel occlusion |
| Stenotic lesions | Area distal to stenosis |
| Initial stages of raised ICP | Advanced stages of raised ICP |
| Feeder vessels of Arteriovenous malformations | |
| Collateral vessels | |
| Sickle cell anemia |
Pulsatility Index
The PI, or Gosling’s PI, is the difference between PSV and EDV divided by MFV. It reflects the cerebrovascular resistance in the distal vascular bed.
PI = (PSV - EDV) / MFV
The PI is inversely proportional to intracranial compliance and CBF. Hence, factors affecting CBF, such as age, sex, PaCO2, and arterial blood pressure, also affect PI. Normal values of PI range from 0.5 to 1.19 (Figure 10). Values of PI less than 0.5 are seen in conditions like arteriovenous malformations, stenosis in the proximal vessels, and hypercapnia. Values of PI more than 1.2 are seen in conditions like raised ICP, stenosis in the distal vessels, and hypocapnia. Although an increase in PI has been used as an indirect measure of raised ICP, one has to remember that it is not specific due to various confounders (Figure 11).8
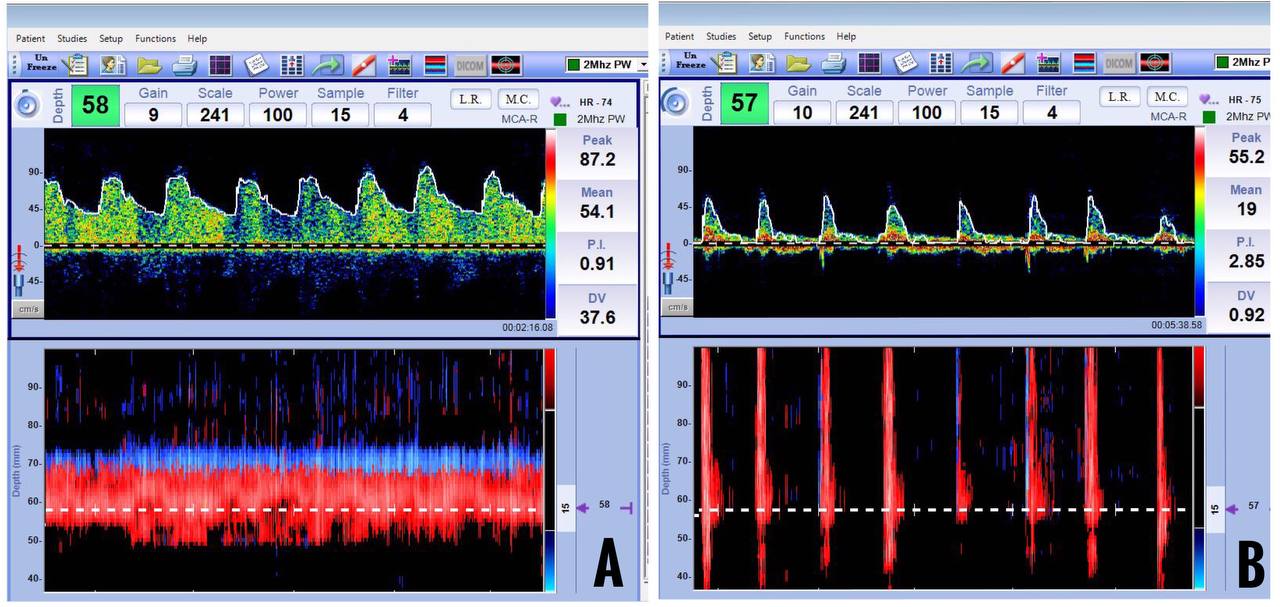
Figure 10: Normal (A) and high (B) Pulsatility Index spectral waveforms
Resistive index
The RI, or Pourcelot’s index, is the difference between PSV and EDV divided by PSV.8 The normal range of RI is 0.49 to 0.63. Like the PI, the RI is also a measure of cerebrovascular resistance in the distal vascular bed. Higher values of RI (more than 0.8) are seen in raised ICP and hydrocephalus. Lower values of RI are seen in hyperemia and vasospasm.
RI = (PSV - EDV) /PSV
Lindegaard Ratio
The LR is the ratio between MFV in the ipsilateral MCA and ipsilateral extracranial ICA. It has been developed to diagnose vasospasm in MCA territory efficiently compared to the velocity criteria alone. Also, the ratio aims to differentiate elevated MFV due to increased flow volume and vasospasm. The normal values of the LR range from 1.2 to 2.5.9 In the event of increased MFV (more than 120 cm/sec) in the MCA territory, the LR of more than three favors the diagnosis of vasospasm over hyperemia. A value of LR more than six indicates severe vasospasm.
LR = MFVMCA / MFVextracranial ICA
Modified Lindegaard Ratio
A modified LR has been developed by Soustiel et al. for basilar artery vasospasm. It is a ratio between MFV in the BA and extracranial VA. A modified LR of more than two is suggestive of basilar artery vasospasm. A ratio of more than three indicates hemodynamically significant basilar artery stenosis.10
Modified LR = MFVBA / MFVextracranial VA
Cerebral Autoregulation
Cerebral autoregulation is the ability of the cerebral vasculature to maintain constant CBF over a wide range of cerebral perfusion pressure (50 - 150 mm Hg). TCD, a non-invasive technique for measuring CBF velocities, is among many methods used to assess cerebral autoregulation. The possibility of continuous and repeated measurements of CBF velocities is the primary advantage of TCD. Methods of evaluation of cerebral autoregulation are broadly divided into two types.
Static Autoregulation
Static Autoregulation is the evaluation of cerebral autoregulation under steady-state conditions. It involves measuring CBF at the baseline and after completion of autoregulatory response secondary to manipulation of arterial blood pressure. Evaluating steady-state responses gives us an idea about the overall efficacy of autoregulatory mechanisms. Tiecks et al. measured static autoregulation using a hypertensive protocol with phenylephrine infusions.11 Static autoregulation is measured as a ratio of the percentage change in estimated cerebrovascular resistance to the percentage change in mean blood pressure. Static Autoregulation is considered intact when the percentage change in estimated cerebrovascular resistance is consistent with the percentage change in mean blood pressure.
Static Autoregulation = (CVR2 - CVR1 / CVR1) / (ABP2 - ABP1 / ABP1) × 100
Where CVR 1 - Cerebrovascular Resistance at baseline
CVR 2 - Cerebrovascular Resistance after autoregulation is achieved
ABP 1 - Mean blood pressure at baseline
ABP 2 - Mean blood pressure achieved after Phenylephrine infusion
Dynamic Autoregulation
Dynamic autoregulation evaluates the characteristic transient response of CBF velocity to a standardized provoked sudden change in blood pressure. However, new and advanced methods do not require provocation measures. Dynamic methods reflect the latency and overall efficacy of cerebral autoregulation. An essential requirement for testing is continuous monitoring of CBF velocity and blood pressure through TCD and hemodynamic monitors, respectively. Another important prerequisite for testing dynamic autoregulation is to have other parameters affecting CBF velocity, such as the PaCO2, constant throughout the procedure. Various methods of testing dynamic autoregulation are listed in Table 5.
| Assessment with Provocative Measures | Assessment without Provocative Measures |
| Thigh cuff release test | Transfer function analysis |
| Assessment with Valsalva maneuver | Assessment of mean velocity index (Mx) |
| Transient hyperemic response test | Assessment of pressure reactivity index (PRx) |
| Assessment with synchronized breathing | |
| Assessment with postural changes |
Transient Hyperemic Response Test
The transient hyperemic response test is used to measure dynamic autoregulation. It was introduced by Giller and standardized by Smielewski et al. Assessment includes obtaining PSV at ipsilateral MCA through TCD before and after compressing the ipsilateral CCA for 5 seconds. An increase in PSV ≥ 10% from baseline is considered intact cerebral autoregulation. The transient hyperaemic response ratio (THRR) can be calculated as well. It is the ratio of PSV after the compression release to PSV before compression. Normal values of THRR in healthy volunteers are 1.105 - 1.29.12 Contraindications to the procedure are carotid artery disease and hemodynamic instability.
THRR = PSV after the compression release / PSV before compression
Cerebral Vasomotor Reactivity
Vasomotor Reactivity (VMR) is the ability of the cerebral vasculature to vasodilate in response to an increase in PaCO2 or a decrease in extracellular pH. It is an indirect measure of cerebrovascular reserve.
Various methods to assess VMR include:
- Breath hold or apnea test
- Carbon dioxide inhalation test
- Diamox test
- Leg cuff method
- L-arginine test
Breath Hold Test
Assessment includes obtaining baseline MFVs at bilateral MCA, holding breath for 30 seconds, and recording MFVs at bilateral MCA from the last 4 seconds of apnea. The breath-holding index (BHI) can be derived by multiplying the ratio of the difference in the MFVs and apnea time by 100. Normal values of BHI are more than 0.6 (Figure 12). Values of BHI between 0.21 and 0.60 imply impaired VMR.13,14
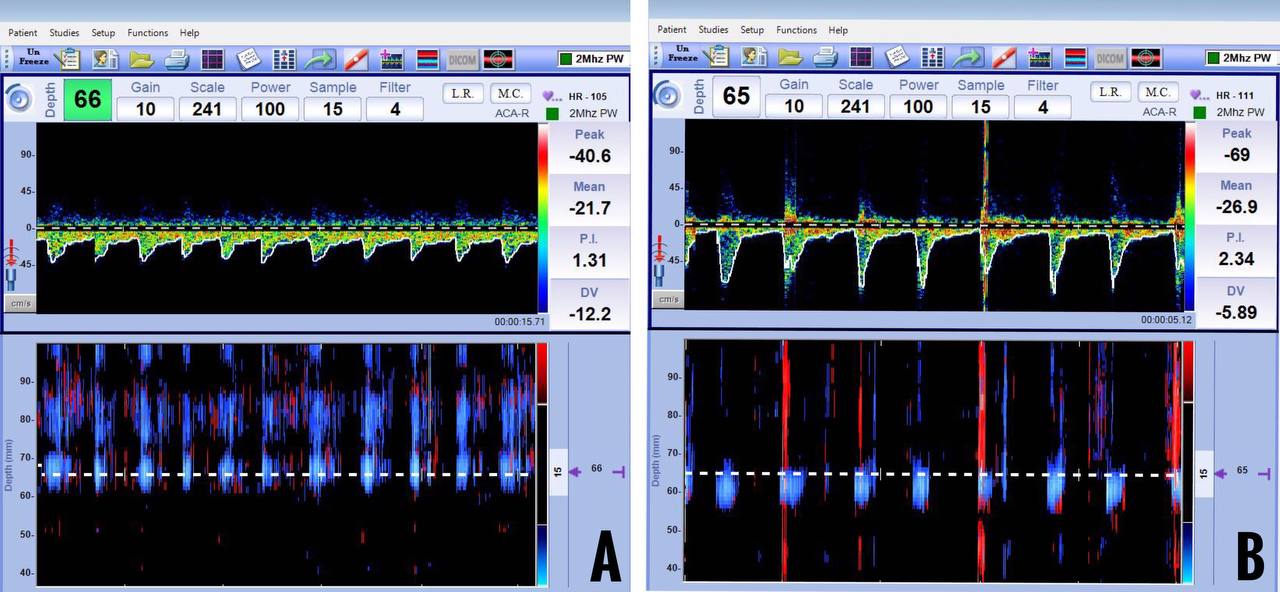
Figure 11: Increased Pulsatility Index (PI) in hypotension with impaired cerebral autoregulation (A) and raised intracranial pressure (B).
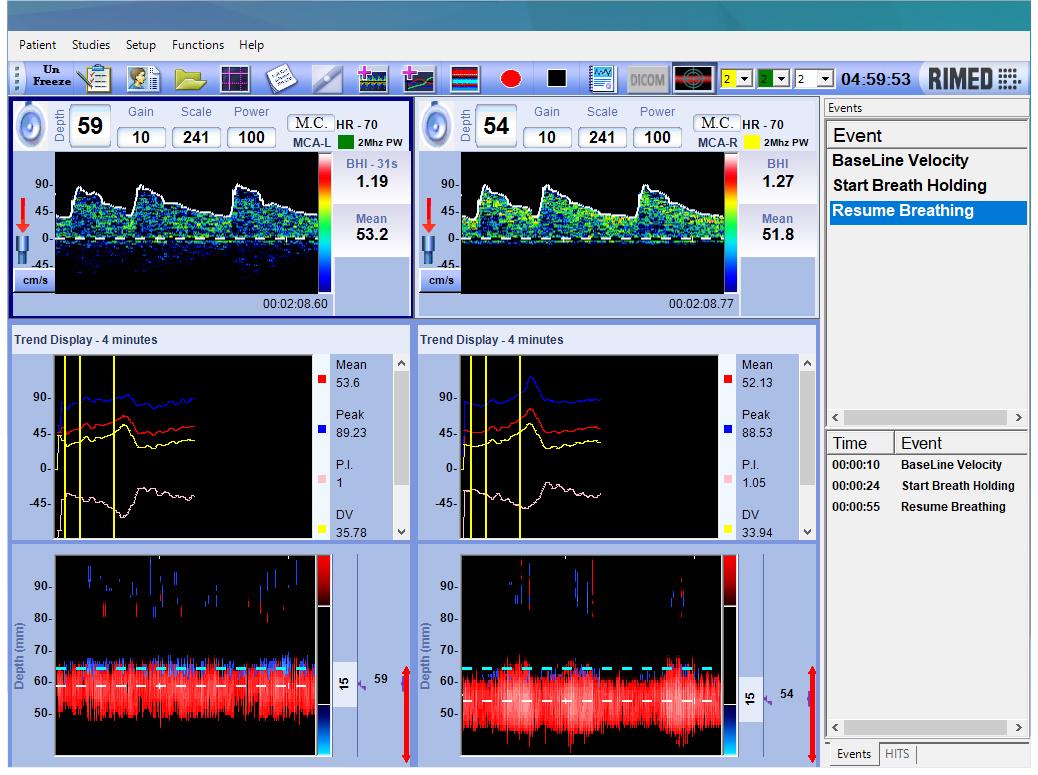
BHI = (MFV maximum – MFV baseline) ÷Apnea time × 100
where MFVbaseline - MFV before starting breath-holding
MFVmax - MFV in the last 4 seconds of breath-holding
Subarachnoid Hemorrhage
The rupture of a cerebral aneurysm is one of the most common causes of subarachnoid hemorrhage (SAH). Among various complications due to SAH, delayed cerebral ischemia (DCI) is an important reason for morbidity and mortality. It occurs in 20%-50% of patients with aneurysmal SAH. Vasospasm is one of the multiple mechanisms that drive the pathogenesis of DCI. Angiographic evidence of vasospasm is seen in up to 70% of the patients after aneurysmal rupture.15 Hemodynamic effects of vasospasm are increased blood velocities in response to the segmental narrowing of blood vessels. The TCD helps in the diagnosis of vasospasm by picking up increased velocities in the spastic blood vessel. Since TCD is a non-invasive and portable test, the temporal evolution of vasospasm can be monitored, and appropriate interventions can be planned.
Velocity criteria for vasospasm in MCA have been well established. An MFV of more than 120 cm/sec suggests vasospasm in the clinical context of aneurysmal SAH. Severe vasospasm is suspected when MFV is more than 200 cm/sec. Sometimes, only a gradual increase in flow velocities per day (more than 50 cm/sec) is observed in the first few days after aneurysmal rupture. One important differential diagnosis for increased velocities is hyperemia. It is important to look at velocities in extracranial circulation to differentiate vasospasm and hyperemia reliably. The LR has been developed specifically for this purpose. A MFV of more than 120 cm/sec and a LR of more than 3 indicate vasospasm (Figure 13).
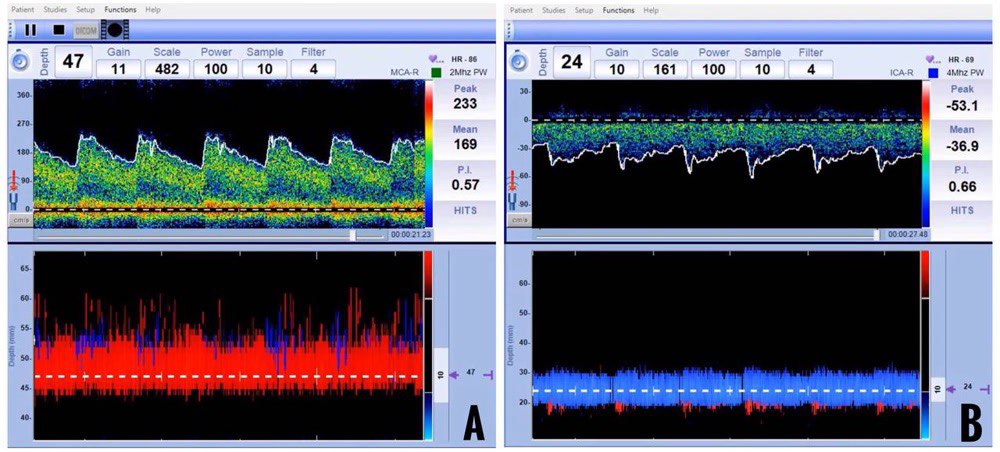
Figure 13: Elevated mean flow velocities in the middle cerebral artery (A) and normal mean flow velocities in the extracranial internal carotid artery (B). The calculated Lindegaard Ratio is 4.6 and suggestive of mild-moderate vasospasm.
A MFV of more than 80 cm/sec and 95 cm/sec suggest vasospasm in vertebral and basilar arteries, respectively. A modified LR of more than two differentiates hyperemia and vasospasm in the basilar artery. The velocities of ACA and PCA have not been standardized because of collateral flows through ACOM and PCOM, respectively.
The Neurocritical Care Society and the European Society of Intensive Care Medicine strongly recommend TCD to predict angiographic vasospasm after aneurysmal SAH.16 The American Heart Association and the American Stroke Association recommend that TCD monitoring is reasonable for detecting vasospasm and predicting DCI in patients with aneurysmal SAH.17 Nevertheless, some limitations exist, such as being operator dependent, difficulty in picking up vasospasm in distal vessels, and difficulty in interpretation of velocities in case of loss of cerebral autoregulation.
Ischemic Stroke
Ischemic stroke is the leading neurological cause of morbidity and mortality in the world. The most common cause of ischemic stroke is a thromboembolic phenomenon due to atherosclerosis of the carotid and intracranial arteries. The role of TCD in managing ischemic stroke includes detecting stenosis or occlusion of intracranial arteries, evaluating collaterals, arterial recanalization after IV thrombolysis, microemboli detection, and prognostication.
The hemodynamic effects of stenosis of large intracranial arteries are focal increase in PSV and MFV. Upstream effects of such lesions include a decrease in MFV and an increase in PI. Downstream effects of such lesions cause dampening of blood flow. The absence of flow signals despite a good insonation window suggests vessel occlusion. Assessment of cerebral autoregulation or VMR status gives additional information regarding the prognosis of ischemic stroke.
Collaterals are vital in maintaining adequate CBF in case of long-standing vessel stenosis or occlusion.18 It has been seen that stroke outcome can be predicted by correlating the degree of collateral circulation with infarct volume and functional status.19 Bedside evaluation of the collateral pathways by TCD is a reliable tool for carotid artery disease. Collaterals open up in case of flow-limiting lesions due to pressure gradient between two anastomosing arterial systems. The important collaterals are listed in Table 6.
| Collateral | Channel | TCD findings |
| Anterior communicating artery | Bilateral cerebral hemispheres |
|
| Posterior communicating artery | Anterior and posterior circulation |
|
| Ophthalmic artery | Extracranial ECA to intracranial ICA |
|
ACA = anterior cerebral artery, ECA = external carotid artery, ICA = internal carotid artery, MFV = mean flow velocity, OA = ophthalmic artery, PCOM = posterior communicating artery, SD = spectral Doppler
Once an ischemic stroke is identified in the window period and thrombolysis is performed, recanalization of the occluded artery is an expected outcome. Recanalization can be assessed by TCD by the reappearance of flow in the vessel or an improvement in the flow with or without reduction in the PI in the proximal segments of the vessel. Demchuk and colleagues have proposed thrombolysis in brain ischemia (TIBI) criteria for the TCD-based classification of MCA flows before and after thrombolysis (Table 7 and Figure 14).
| TIBI SCORE | FLOW STATUS IN MCA | TCD FINDINGS |
| 0 | Absent | Absence of regular pulsatile flow signals |
| 1 | Minimal |
|
| 2 | Blunted |
|
| 3 | Dampened |
|
| 4 | Stenotic |
|
| 5 | Normal |
|
EDV = end diastolic velocity, MFV = mean flow velocity, P = pulsatility index, TIBI Score = thrombolysis in brain ischemia score

Figure 14: Thrombolysis in brain ischemia (TIBI) score [Image adapted from Kargiotis et al.]25
Detection of Transient Microemboli
Microemboli originating from various pathologies can reach the cerebral circulation and cause stroke. Common sources of microemboli include atherosclerotic lesions in the carotid artery, cardiac thrombus, venous thrombus entering systemic circulation through a right to left shunt, and interventional procedures such as angioplasty. Among various investigations, TCD is the only imaging modality used for real-time detection of microemboli entering cerebral circulation. Microemboli traveling in the blood vessels are captured by TCD as high-intensity transient signals (HITS) or microembolic signals (MES).
Characteristics of HITS/ MES on spectral waveform are (Figure 15)21:
- Duration less than 300 ms
- Amplitude > 3 dB higher than the background blood flow signal
- Unidirectional within the Doppler velocity spectrum
- Audio signals resembling a snap, a chirp, or a moan
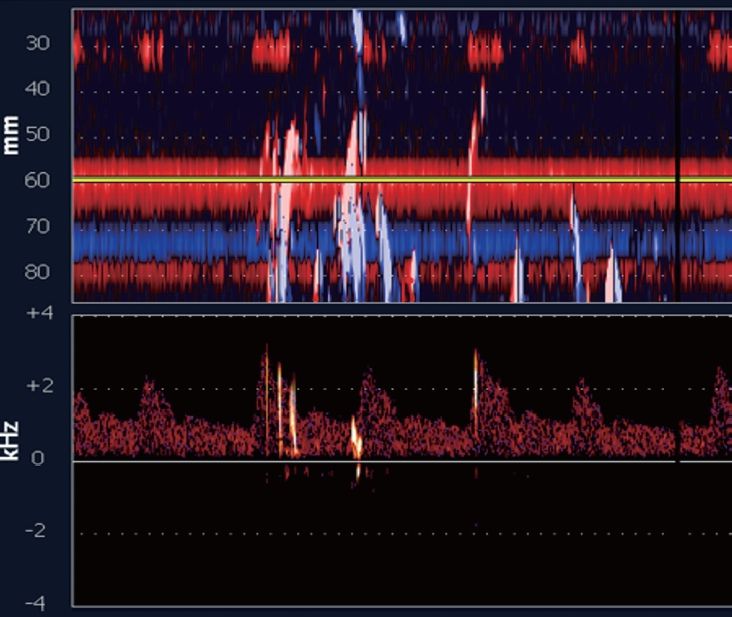
Image adapted from Malhotra et al.26
Assessment of microemboli secondary to right to left shunt includes:
- The patient should be placed supine. Hemodynamic monitors and TCD equipment are attached.
- Intravenous contrast agents, such as agitated saline, are injected into the cubital vein through an 18 gauge cannula to mimic the microemboli phenomenon.
- Provocation measures, most commonly a Valsalva maneuver, are performed to improve the sensitivity of microemboli detection in case of an initial negative test.
- Ideally, the MCA on both sides is simultaneously insonated by TCD. In case of difficulty, the MCA on one side can be insonated for assessment.
- Audiovisual assessment of spectral waveform is a must.
- The counting of HITS/MES should be manually verified even if there is an automated detection.
- Even a single HITS/MES captured on TCD is considered positive for the presence of a shunt. The number of HITS/MES detected will help grade the shunt (Table 8).22
- Keeping an eye on the timing of the appearance of HITS/MES helps differentiate the location of the shunt as cardiac and extracardiac.
| Grade of Shunt | Number of HITS/MES on TCD spectrum - Unilateral Monitoring | Number of HITS/MES on TCD spectrum - Bilateral Monitoring |
| Grade 0 | 0 | 0 |
| Grade 1 | 1-10 | 1-20 |
| Grade 2 | > 10 but no curtain effect | > 20 but no curtain effect |
| Grade 3 | Curtain effect (uncountable HITS/MES) | Curtain effect (uncountable HITS/MES) |
TCD = transcranial Doppler, HITS = high-intensity transient signals, MES = microemboli signals
Microemboli secondary to pathologies other than shunt can be assessed without IV contrast agents and provocative measures. One important limitation of TCD assessment of microemboli is that it cannot localize and define the source of microemboli. Also, robust criteria do not exist to differentiate gaseous and solid microemboli.
Cerebral Circulatory Arrest
Cerebral Circulatory Arrest (CCA) is an outcome of uncontrolled intracranial hypertension secondary to any form of acute brain injury. Global ischemia caused by CCA is the most common pathway leading to brain death. Brain death is a clinical condition in which the functions of the entire brain, including the brainstem, are irreversibly cessated.
An increase in ICP causes elevated cerebrovascular resistance in the distal blood vessels. This manifests as an initial increase in PSV, a decrease in EDV, and an increase in the PI in the TCD. EDV reaches baseline when ICP equals diastolic pressure. In case of a further increase in the ICP equalling systolic pressure, oscillating flow is observed, wherein forward and reverse flow components in one cardiac cycle exhibit almost the same area. Systolic spikes are seen when ICP is more than systolic pressure, which is highly suggestive of cerebral circulatory arrest when present bilaterally. They are sharp unidirectional velocity signals in early systole of less than 200 ms duration, less than 50 cm/sec, and without a flow signal during the remaining cardiac cycle. Gradually, systolic spikes will disappear, and no flow signals can be observed (Figure 16).
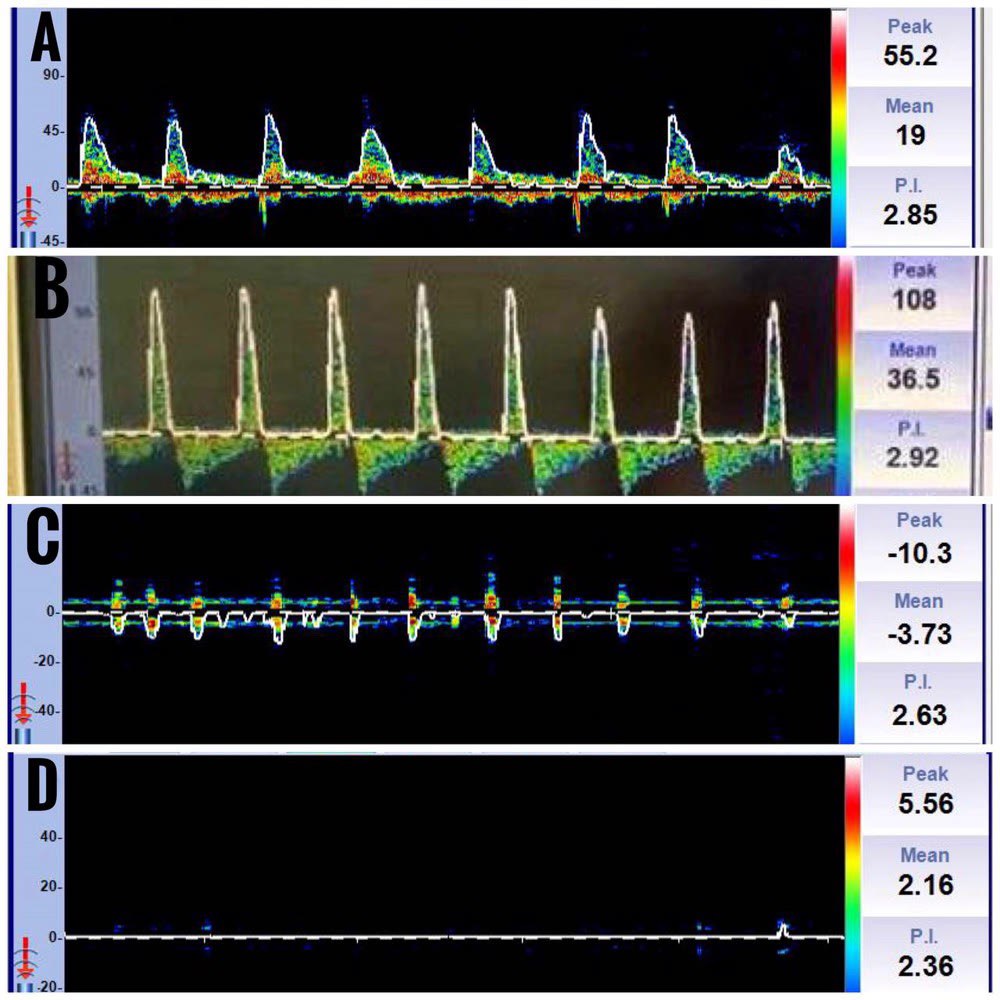
(A) Decrease in mean flow velocity and end-diastolic velocity, (B) Diastolic flow reversal, (C) Waveforms resembling systolic spikes, (D) No flow state
The TCD is used as an ancillary test for the assessment of CCA. It is initiated when clinical examination suggests the absence of cerebral and brainstem functions. TCD findings are suggestive of cerebral circulatory arrest when:23
- Bilateral MCA insonation reveals an oscillating pattern, systolic spike, or absent flow signals.
- Bilateral extracranial ICA insonation reveals an oscillating pattern.
- Similar findings are obtained in the second examination done at an interval of at least 30 minutes.
American Academy of Neurology guidelines on brain death/death by neurological criteria in adults issued in 2023 recommend that clinicians use TCD as an ancillary test to aid the diagnosis of brain death/death by neurological criteria in adults.24
Summary
TCD is a rapid, inexpensive bedside tool that can be used along with a battery of other tests in the clinical diagnosis and management of several cerebrovascular disorders, such as acute ischemic stroke, subarachnoid hemorrhage, and traumatic brain injury. It is also helpful in detecting collateral flow, managing cerebrovascular atherosclerotic diseases, and confirming brain death. It has valuable application in different settings to study cerebral autoregulation and vasomotor reactivity in healthy and diseased populations.
We would like to acknowledge support from Saurabh Anand, MD, chief of neuroanaesthesia and neurocritical care, Artemis Hospital, Gurugram, India; Kanika Gupta, MD, academic senior resident, department of anaesthesiology, AIIMS, Rishikesh, India; and Mayank Gaur, director, Indresh Sales and Services, Delhi, India, in providing us with important images.




References:
- Aaslid R, Markwalder TM, Nornes H. Noninvasive transcranial Doppler ultrasound recording of flow velocity in basal cerebral arteries. J Neurosurg 1982;57(6):769-74. https://doi.org/10.3171/jns.1982.57.6.0769
- Moehring MA, Spencer MP. Power M-mode Doppler (PMD) for observing cerebral blood flow and tracking emboli. Ultrasound Med Biol 2002;28(1):49-57. https://doi.org/10.1016/s0301-5629(01)00486-0
- Ringelstein EB, Kahlscheuer B, Niggemeyer E, et al. Transcranial Doppler sonography: anatomical landmarks and normal velocity values. Ultrasound Med Biol 1990;16(8):745-61. https://doi.org/10.1016/0301-5629(90)90039-f
- Alexandrov AV, Sloan MA, Wong LKS, et al. for the American Society of Neuroimaging Practice Guidelines Committee. Practice standards for transcranial Doppler ultrasound: Part I—test performance. J Neuroimaging 2007;17:11-8. https://doi.org/10.1111/j.1552-6569.2006.00088.x
- Purkayastha S, Sorond F. Transcranial Doppler ultrasound: technique and application. Semin Neurol 2012;32(4):411-20. https://doi.org/10.1055/s-0032-1331812
- Nicoletto HA, Burkman MH. Transcranial Doppler series part III: interpretation. Am J Electroneurodiagnostic Technol 2009;49(3):244-59
- Blanco P, Abdo-Cuza A. Transcranial Doppler ultrasound in neurocritical care. J Ultrasound 2018;21(1):1-16. https://doi.org/10.1007/s40477-018-0282-9
- Naqvi J, Yap KH, Ahmad G, et al. Transcranial Doppler ultrasound: a review of the physical principles and major applications in critical care. Int J Vasc Med 2013;2013:629378. https://doi.org/10.1155/2013/629378
- Lindegaard KF, Nornes H, Bakke SJ, et al. Cerebral vasospasm diagnosis by means of angiography and blood velocity measurements. Acta Neurochir (Wien) 1989;100(1-2):12-24. https://doi.org/10.1007/BF01405268
- Soustiel JF, Shik V, Shreiber R, et al. Basilar vasospasm diagnosis: investigation of a modified "Lindegaard Index'' based on imaging studies and blood velocity measurements of the basilar artery. Stroke 2002;33(1):72-7. https://doi.org/10.1161/hs0102.100484
- Tiecks FP, Lam AM, Aaslid R, et al. Comparison of static and dynamic cerebral autoregulation measurements. Stroke 1995;26(6):1014-9. https://doi.org/10.1161/01.str.26.6.1014
- Smielewski P, Czosnyka M, Kirkpatrick P, et al. Assessment of cerebral autoregulation using carotid artery compression. Stroke 1996;27(12):2197-203. https://doi.org/10.1161/01.str.27.12.2197
- Nicoletto HA, Boland LS. Transcranial Doppler series part v: specialty applications. Am J Electroneurodiagnostic Technol 2011;51(1):31-41.
- Markus HS, Harrison MJ. Estimation of cerebrovascular reactivity using transcranial Doppler, including the use of breath-holding as the vasodilatory stimulus. Stroke 1992;23(5):668-73. https://doi.org/10.1161/01.str.23.5.668
- Chalet FX, Briasoulis O, Manalastas EJ, et al. Clinical burden of angiographic vasospasm and its complications after aneurysmal subarachnoid hemorrhage: a systematic review. Neurol Ther 2023;12(2):371-90. https://doi.org/10.1007/s40120-022-00436-7
- Le Roux P, Menon DK, Citerio G, et al. Consensus summary statement of the International Multidisciplinary Consensus Conference on Multimodality Monitoring in Neurocritical Care: a statement for healthcare professionals from the Neurocritical Care Society and the European Society of Intensive Care Medicine. Intensive Care Med 2014;40(9):1189-1209. https://doi.org/10.1007/s00134-014-3369-6
- Hoh BL, Ko NU, Amin-Hanjani S, et al. 2023 Guideline for the Management of Patients With Aneurysmal Subarachnoid Hemorrhage: A Guideline From the American Heart Association/American Stroke Association. Stroke 2023;54(7):e314-e70. https://doi.org/10.1161/STR.0000000000000436. Erratum in: Stroke 2023;54(12):e516. https://doi.org/10.1161/STR.0000000000000449
- Liebeskind DS. Understanding blood flow: the other side of an acute arterial occlusion. Int J Stroke 2007;2(2):118-20. https://doi.org/10.1111/j.1747-4949.2007.00117.x
- Lau AY, Wong EH, Wong A, et al. Significance of good collateral compensation in symptomatic intracranial atherosclerosis. Cerebrovasc Dis 2012;33(6):517-24. https://doi.org/10.1159/000337332
- Demchuk AM, Burgin WS, Christou I, et al. Thrombolysis in brain ischemia (TIBI) transcranial Doppler flow grades predict clinical severity, early recovery, and mortality in patients treated with intravenous tissue plasminogen activator. Stroke 2001;32(1):89-93. https://doi.org/10.1161/01.str.32.1.89
- Consensus Committee of the Ninth International Cerebral Hemodynamic Symposium. Basic identification criteria of Doppler microembolic signals. Stroke 1995;26:1123.
- Jauss M, Zanette E. Detection of right-to-left shunt with ultrasound contrast agent and transcranial Doppler sonography. Cerebrovasc Dis 2000;10(6):490-6. https://doi.org/10.1159/000016119
- Ducrocq X, Hassler W, Moritake K, et al. Consensus opinion on diagnosis of cerebral circulatory arrest using Doppler-sonography: Task Force Group on cerebral death of the Neurosonology Research Group of the World Federation of Neurology. J Neurol Sci 1998;159(2):145-50. https://doi.org/10.1016/s0022-510x(98)00158-0
- Greer DM, Kirschen MP, Lewis A, et al. Pediatric and adult brain death/death by neurologic criteria consensus guideline. Neurology 2023;101(24):1112-32. https://doi.org/10.1212/WNL.0000000000207740. Erratum in: Neurology 2024 13;102(3):e208108. https://doi.org/10.1212/WNL.0000000000208108
- Kargiotis O, Psychogios K, Safouris A, et al. Transcranial Doppler monitoring of acute reperfusion therapies in acute ischemic stroke patients with underlying large vessel occlusions. J Neurosonol Neuroimag 2020;12(1):10-25. https://doi.org/10.31728/jnn.2020.00084
- Malhotra K, Katsanos AH, Frogoudaki A, et al. Patent foramen ovale: risk factors and diagnostic comparison of transcranial doppler versus echocardiography for secondary stroke prevention. J Neurosonol Neuroimag 2019;11(1):22-33. https://doi.org/10.31728/jnn.2019.00042