How I Do It: Ultrasound-Guided Thoracic Paravertebral Block
Cite as: Sivakumar RK, Karmakar MK. How I do it: ultrasound-guided thoracic paravertebral block. ASRA Pain Medicine News 2025;50. https://doi.org/10.52211/asra050125.007.
How I Do It
Introduction
Thoracic paravertebral block (TPVB) involves injecting local anesthetic (LA) alongside the thoracic vertebra, close to where the spinal nerves exit the intervertebral foramen.1,2 This results in ipsilateral, somatic, and sympathetic nerve blockade across multiple contiguous thoracic dermatomes.1,2 Unlike central neuraxial blocks, like epidural3 and spinal anesthesia,4 TPVB can altogether abolish somatosensory evoked potentials from the level of LA injection,5 providing high-quality afferent nociceptive blockade for managing unilateral thoracic and abdominal pain.1 TPVB, combined with intravenous sedation, has also been used to provide surgical anesthesia for primary breast cancer6 and hernia7 surgeries. Bilateral TPVB has also been described.8 TPVB is traditionally performed using surface anatomical landmarks,1 but in recent years, interest in using ultrasound for TPVB has grown, leading to several technical variations.9-12 This article briefly outlines the basic principles of TPVB, sonoanatomy of the thoracic paravertebral space (TPVS), and how the authors perform ultrasound-guided (USG) TPVB.
This article briefly outlines the basic principles of TPVB, sonoanatomy of the thoracic paravertebral space (TPVS), and how the authors perform ultrasound-guided (USG) TPVB.
Brief History
Hugo Sellheim (1871-1936) from Leipzig, Germany, pioneered TPVB in 1905, and the early 1900s, it was used extensively to provide anesthesia and analgesia for abdominal surgery.13,14 Despite its initial popularity, TPVB fell out of favor for unknown reasons until 1979 when Eason and Wyatt revived interest by describing a catheter technique.15 Interest in TPVB resurged in the late 20th century for both adults1 and children.16,17 During this period, our understanding of TPVB improved significantly, thanks to contributions from Sabanathan,2,8; 18-20 Lonnqvist,13,16; 21-24 and Richardson.2,8; 13,14; 18-20 The introduction of two-dimensional ultrasound for regional anesthesia by Kapral and colleagues in 199425 has led to a steady increase in the number of publications on the use of ultrasound for TPVB.12 Hara and colleagues from Japan first described USG TPVB in 2007.26
Functional Anatomy
The TPVS is a wedge-shaped space located on either side of the thoracic vertebral column behind the parietal pleura (Figure 1).1 It is wider on the left than on the right, and the base is formed by the vertebral body, intervertebral disc, and intervertebral foramen (Figure 1).1 The parietal pleura forms the anterolateral boundary (Figures 1-2), while the superior costotransverse ligament (SCTL), which extends from the lower border of the transverse process (TP) above to the upper border of the TP below, forms the posterior boundary (Figures 2-3).1 The inter-transverse ligament, thinner than the SCTL, also extends between adjacent transverse processes (Figure 3). The SCTL is continuous laterally with the internal intercostal membrane (IICM), the medial extension of the internal intercostal muscle (Figure 3). The apex of the TPVS is continuous with the posterior intercostal space (PICS) lateral to the tip of the TP (Figures 1 and 3).1 While the SCTL forms the posterior boundary of the TPVS, it is not a water-tight barrier. Fenestrations and gaps have been identified within the architecture of the SCTL,27-29 and an injection behind the SCTL or into the intertransverse tissue complex (ITTC) can make its way to the paravertebral space.30,31 The ITTC comprises the SCTL, intertransverse ligament, posterior costotransverse ligament (if present), intertransverse and levator costarum muscles, and fatty connective tissue.32
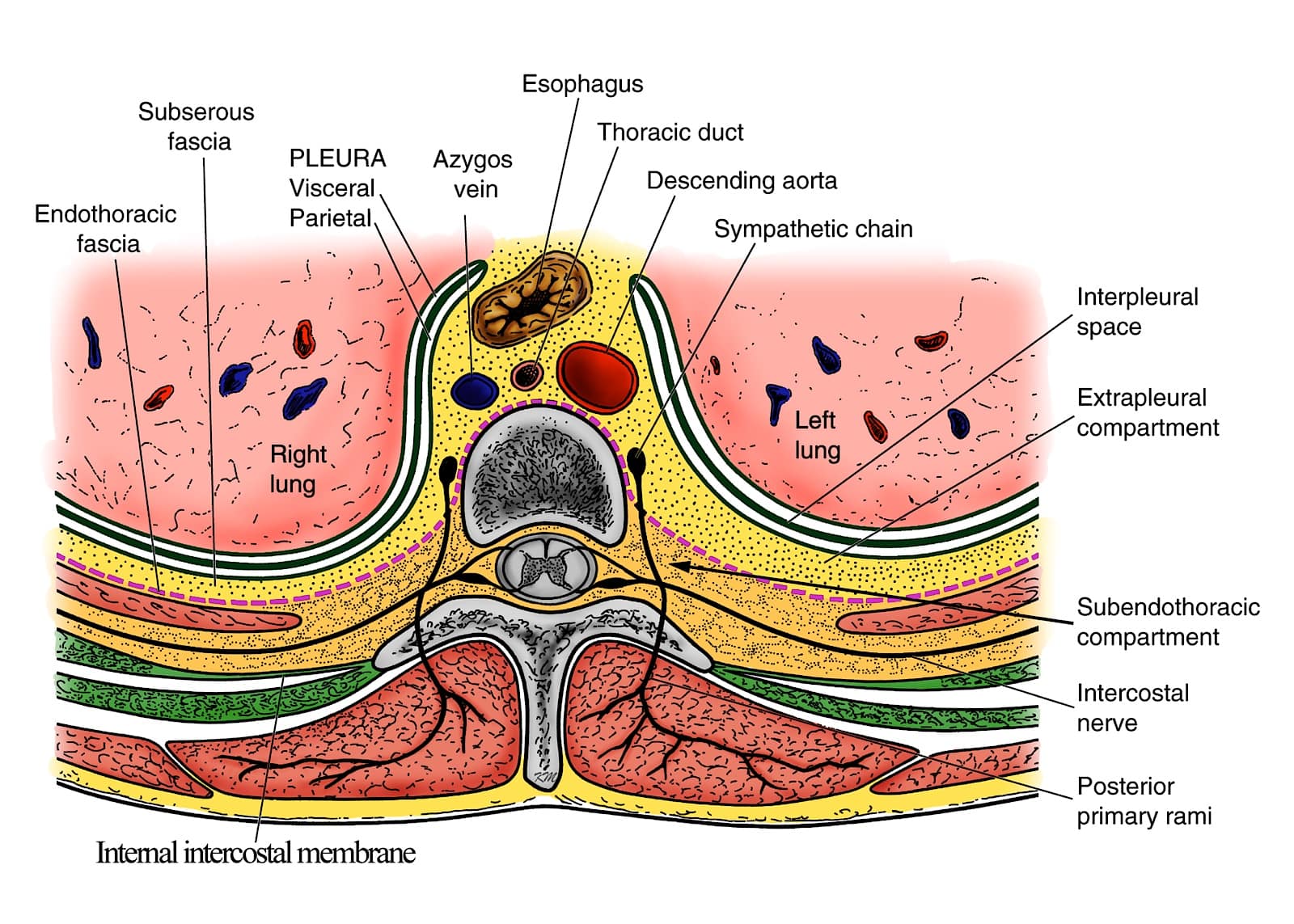
Figure 1. Transverse anatomy of the thoracic paravertebral region.
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
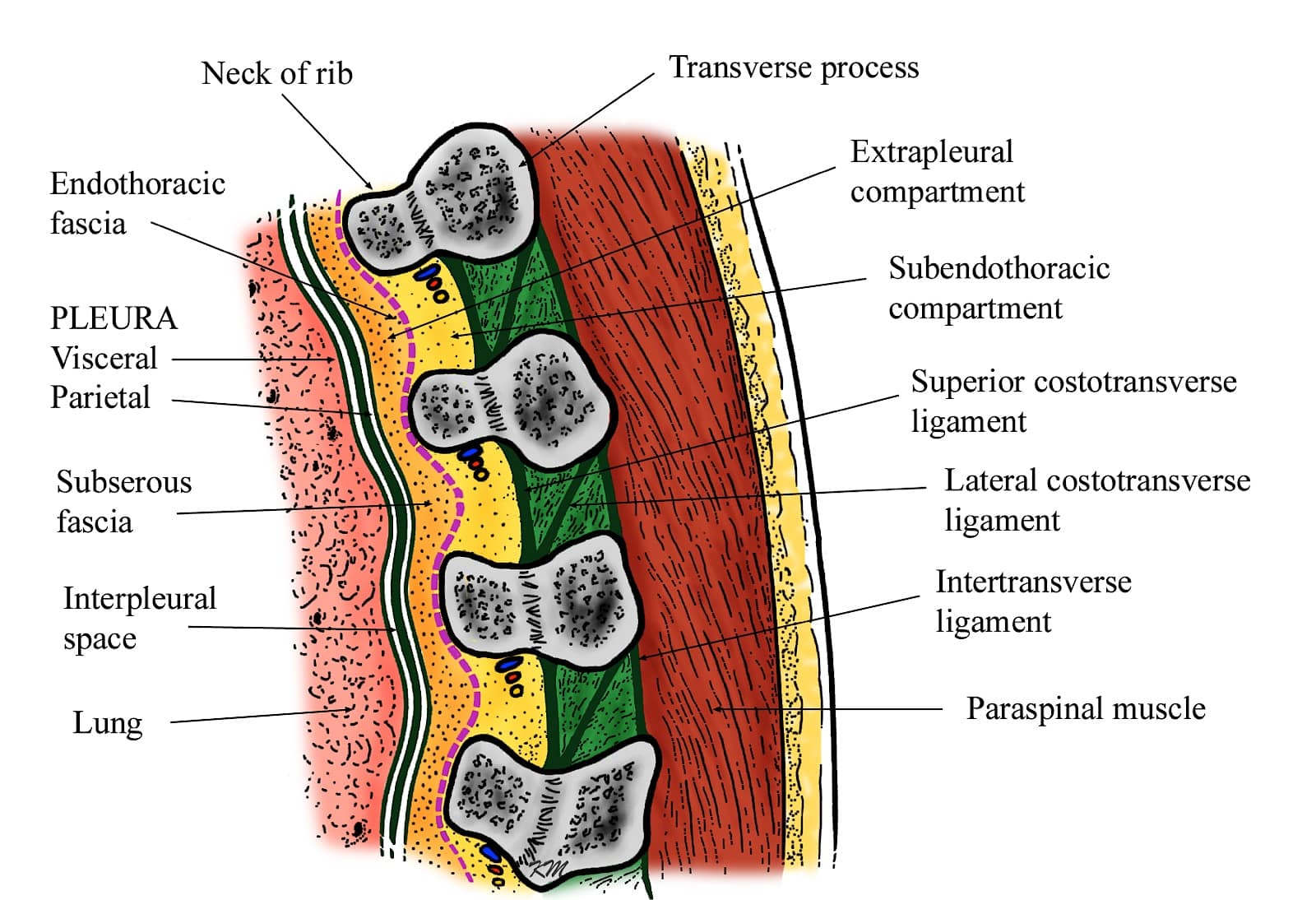
Figure 2. Sagittal anatomy of the thoracic paravertebral region.
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
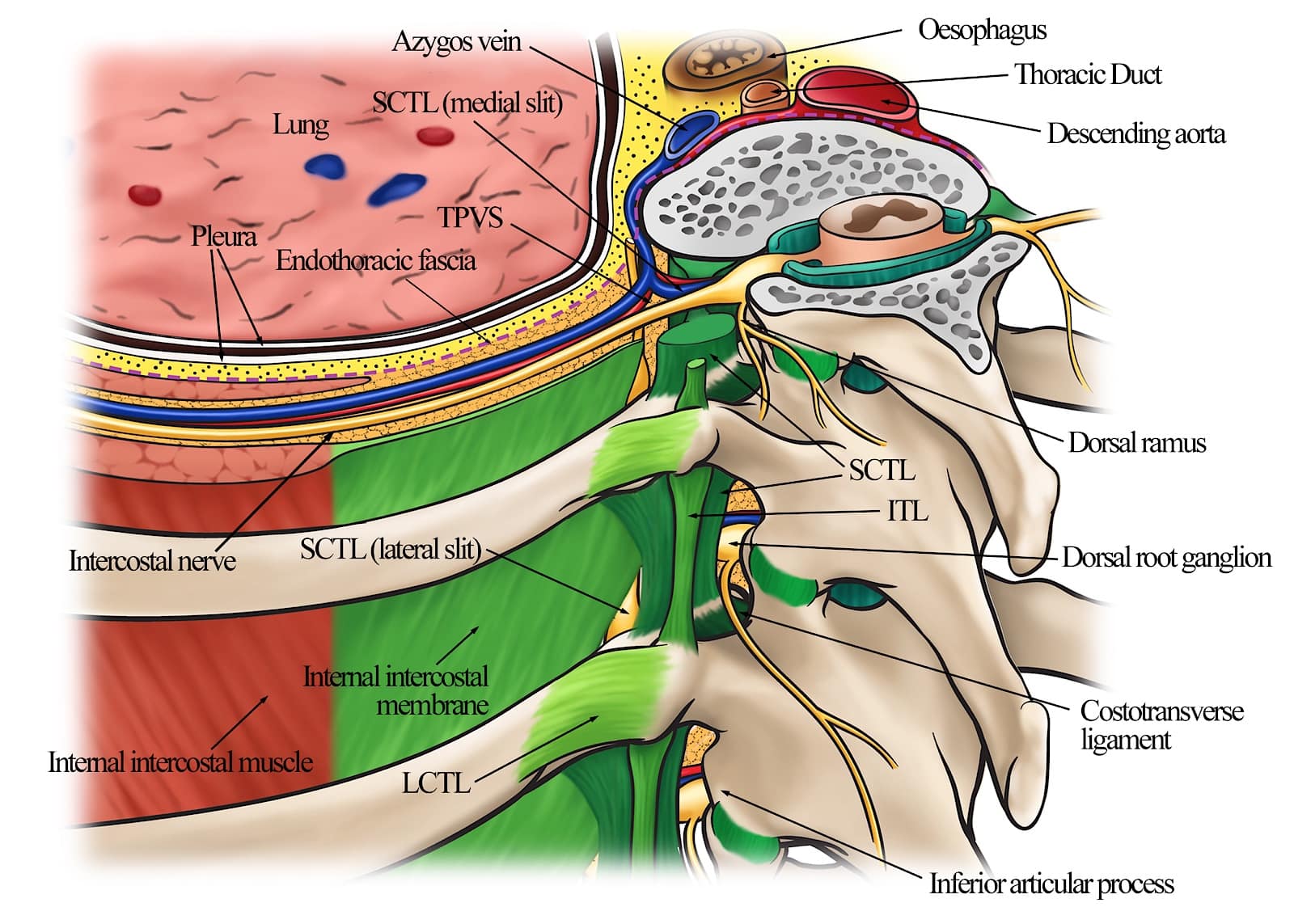
Figure 3. The thoracic paravertebral region's anatomy shows the various paravertebral ligaments attached to the transverse processes of the thoracic vertebra and their anatomic relationship to the thoracic paravertebral space.
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
Between the parietal pleura and the SCTL lies the fibroelastic endothoracic fascia (Figure 1-2), which is the deep fascia of the thorax and lines the internal aspect of the thoracic cage (Figure 4).33,34 At the TPVS, the endothoracic fascia is loosely attached to the ribs (Figure 2) and/or the TP and fuses medially with the periosteum of the vertebral body (Figure 1).1 The subserous fascia, a layer of loose connective tissue, lies between the parietal pleura and the endothoracic fascia (Figure 1).1 The endothoracic fascia divides the TPVS into the anterior extrapleural paravertebral compartment and the posterior subendothoracic paravertebral compartment (Figure 1-2).1 The TPVS contains fatty tissue within which lie the intercostal (spinal) nerve, dorsal ramus, intercostal vessels, rami communicantes, and the sympathetic chain (Figure 1-2).1 The spinal nerve in the TPVS divides into multiple small bundles without a fascial sheath, making them vulnerable to injected LA.1 The intercostal nerve and vessels are located behind the endothoracic fascia, while the sympathetic trunk is located anterior to it (Figure 2-4).1
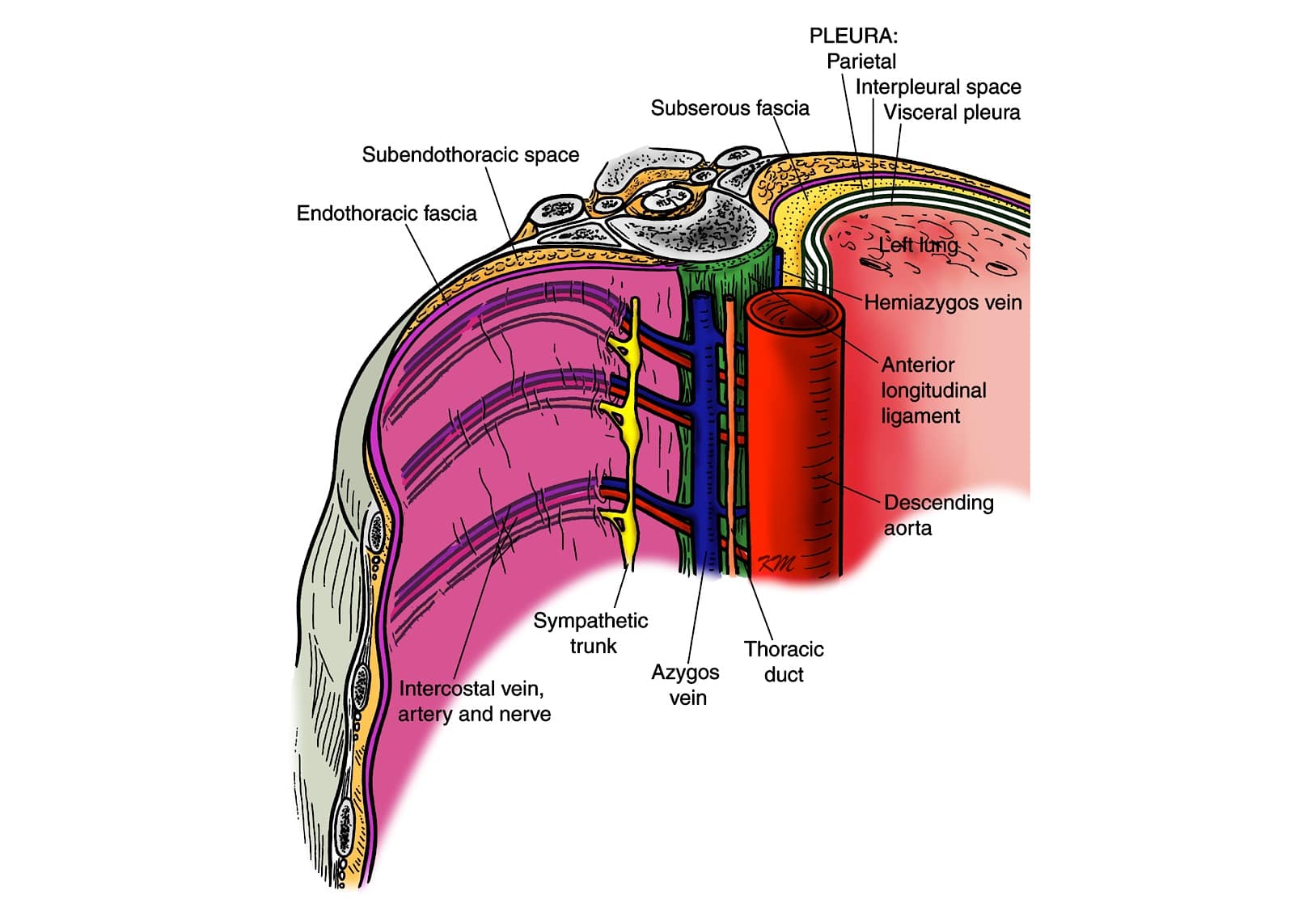
Figure 4. The endothoracic fascia and its anatomic relationship to the thoracic paravertebral space. Note the fascial compartments and the location of the neurovascular structures in relation to the endothoracic fascia.
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
The TPVS is continuous with the contiguous space above and below,34 the epidural space medially via the intervertebral foramen,35 the intercostal space laterally; and the contralateral TPVS via the epidural and prevertebral space (Figure 1-3.).34 Communication with the contiguous paravertebral spaces above and below occurs not only via the paravertebral space,34 but also via the costotransverse space.36-38 The anatomy of the TPVS has recently been redefined with the characterization of the SCTL and identification of the “retro-SCTL space” (Figures 5 and 6), using three-dimensional micro-CT imaging of paravertebral tissue specimens in cadavers.36 Contrary to previous understanding,1 the SCTL forms an incomplete posterior wall of the TPVS with the lateral and medial slits (see Figures 3 and 5).29,39 The former lies laterally between the IICM and the SCTL while the latter lies medially between the SCTL and the vertebral body (Figures 3 and 5).29,39 The costotransverse space, located between the neck of the rib and the transverse process, is filled with fat and loose connective tissue and is visible as a distinct anatomical space in micro-CT images.36 The retro-SCTL space36 also filled with fat, and connective tissue communicates with the erector spinae and retrolaminar planes posteriorly, TPVS anteriorly via the lateral and medial slits in the SCTL,29 intervertebral canal and epidural space medially,37 the intercostal space laterally via the lateral slit in the SCTL (Figures 5 and 6), and with the contiguous TPVS and retro-SCTL space above and below via the costotransverse space.36,37 The location of the costotransverse foramen within this redefined paravertebral anatomy is unclear. However, it might represent the medial end of the retro-SCTL space as depicted in Figures 5 and 6,29,37 given its proximity to the base of the transverse process and inferior articular process (IAP).
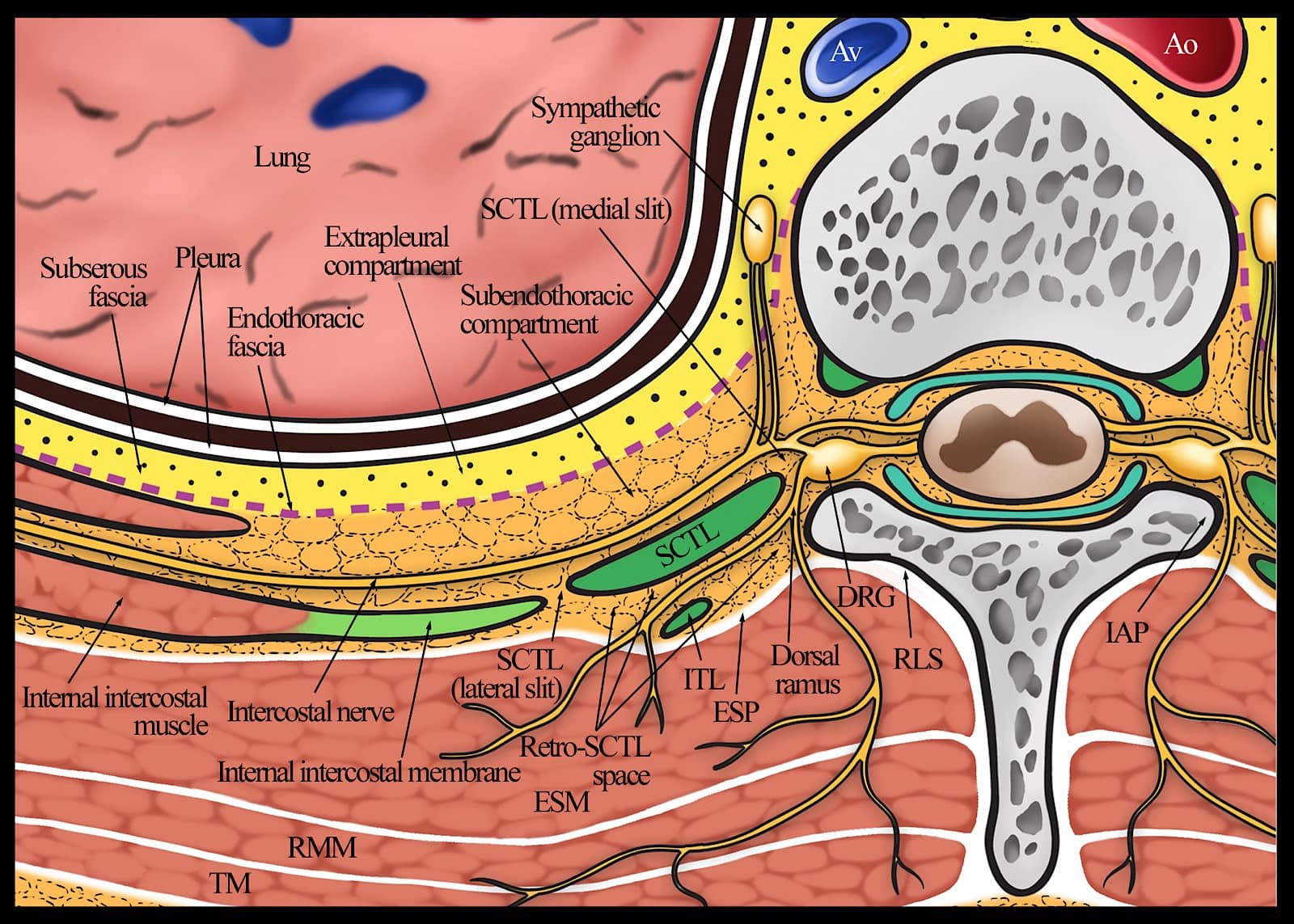
Figure 5. Transverse anatomy of the thoracic paravertebral region showing the anatomic relationship of the thoracic paravertebral space to the retro-SCTL space and neuraxis. Note the medial and lateral slits in the SCTL and the position (not to scale) of the ITL. Also note that both the ventral and dorsal rami of the spinal nerve are initially located within the medial aspect of the retro-SCTL space before the ventral ramus enters the thoracic paravertebral space through the medial slit in the SCTL, and the dorsal rami travels posteriorly toward the paraspinal muscles.
Ao = descending aorta, Av = azygos vein, DRG = dorsal root ganglion, ESP = erector spinae plane, ESM = erector spinae muscle, IAP = inferior articular process, ITL = intertransverse ligament, RLS = retrolaminar space, RMM = rhomboid major muscle, SCTL = superior costotransverse ligament, TM = trapezius muscle
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
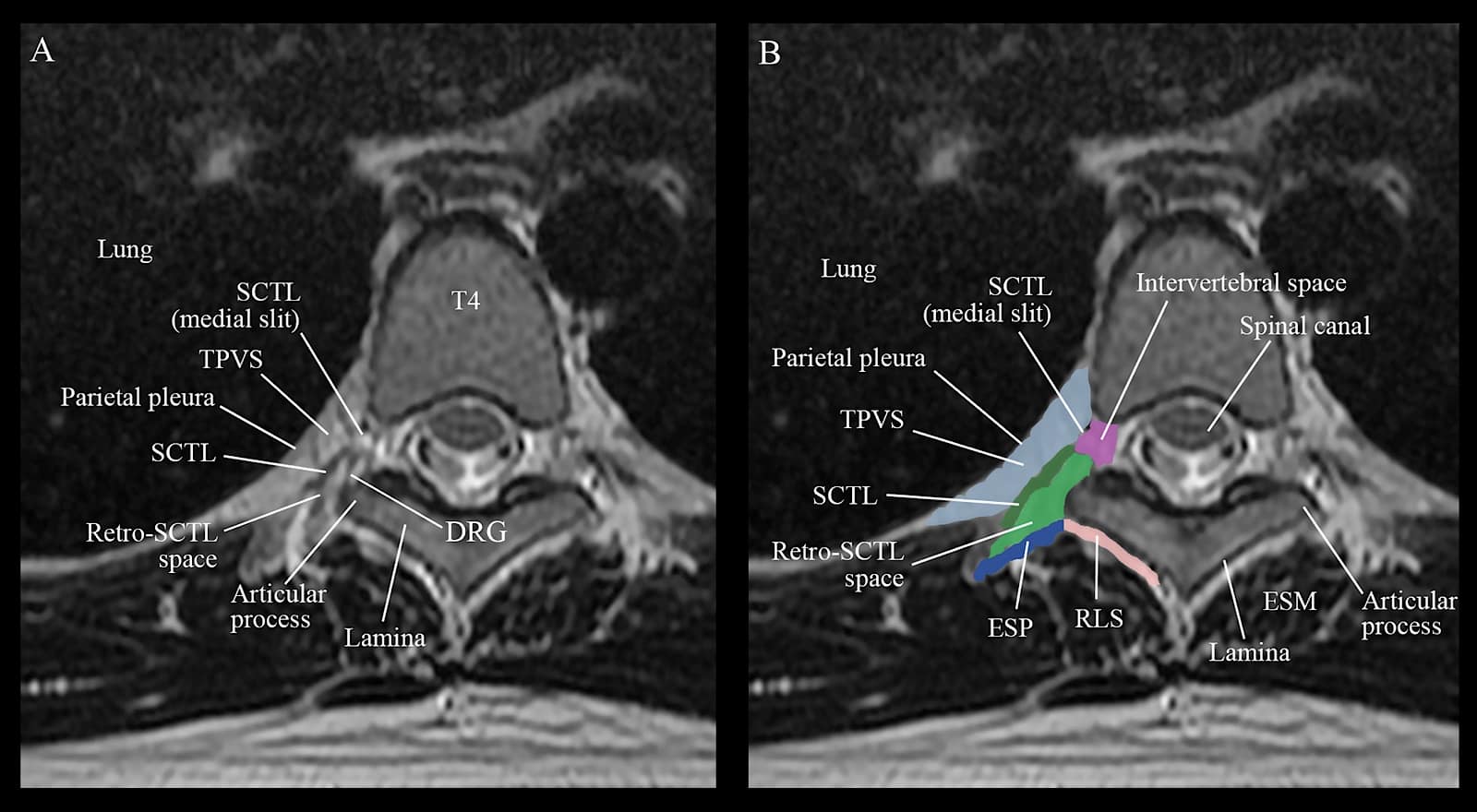
Figure 6. (A) Axial (transverse) MRI of the thoracic paravertebral region from the level of the T4 vertebral body and inferior articular process corresponding to the level at which the authors perform ultrasound-guided thoracic paravertebral block. Note the clear depiction of the superior costotransverse ligament (SCTL), retro-SCTL space, and the potential communications between the fascial planes behind the intertransverse tissue complex and the thoracic paravertebral space (TPVS). Two images of the same MRI are presented, with no color highlights (A) and with color (B) highlights assigned to various anatomic structures and tissue planes for clarity and comparison, using Adobe Photoshop (Adobe, San Jose, California, USA). Note the anatomic relationship of the retro-SCTL space to the ESP (erector spinae plane), retrolaminar space (RLS), TPVS, and the intervertebral space (foramen). DRG represents dorsal root ganglion; ESM, and erector spinae muscle.
SCTL = superior costotransverse ligament, TPVS = thoracic paravertebral space, ESP = erector spinae plane, RLS = retrolaminar space, DRG = dorsal root ganglion, ESM = erector spinae muscle
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
Three-dimensional micro-CT images suggest that, contrary to traditional belief, the ventral ramus of the thoracic spinal nerve doesn’t directly enter the TPVS.36 Both the proximal parts of the ventral and dorsal rami initially lie within the medial retro-SCTL space near the intervertebral foramen.36 The ventral ramus accesses the TPVS through the medial slit in the SCTL or by piercing the ligament.36 In contrast, the dorsal ramus traverses laterally through the fat-filled retro-SCTL space to the erector spinae plane.36 Another smaller ligament identified as the intertransverse ligament is located behind the SCTL (Figures 3 and 5).36 Small branches from the dorsal ramus pass through the interligamentary space between these two ligaments before extending laterally.36
Mechanism of Nerve Blockade
A thoracic paravertebral injection may remain localized to the site of injection, or it may spread to the contiguous paravertebral spaces above and below the site of injection,34 the epidural space medially, the intercostal space laterally,1,35 or a combination of the above to affect ipsilateral somatic2,5 and sympathetic nerves,2 including the posterior primary ramus in multiple contiguous thoracic dermatomes.1,2 The majority of the published data describing paravertebral spread or distribution of anesthesia after a TPVB to date are with landmark-based techniques1,2,40,41 and data describing the segmental spread of anesthesia after a USG TPVB is only just emerging.42-45 Therefore, one must exercise caution interpreting published data on the segmental distribution of anesthesia after a TPVB since it may not apply to a USG TPVB with a different direction of needle advancement (anatomical landmark-based TPVB – posteroanterior needle insertion, while USG TPVB – lateral to medial needle insertion for the transverse in-plane technique described below).
Segmental Dermatomal Anesthesia
Anatomical landmark-based TPVB produces ipsilateral, multi-dermatomal, segmental thoracic anaesthesia.1,2,40,41 Fifteen ml of bupivacaine 0.5% injected into the TPVS produces mean unilateral somatic block of 5 (range 1 – 9) dermatomes and sympathetic block of 8 (range 6 – 10) dermatomes.2 However, the dermatomal distribution of anesthesia is often unpredictable1,2,40,41 and variable. The reason for this variability is not clear, but anatomical and physical factors have been theorized.1,2,40 Factors such as age, gender, height, weight, injectate volume or the dose of bupivacaine, previous posterolateral thoracotomy, or the spread of a radio-opaque contrast medium do not appear to affect the distribution of sensory blockade after a single-injection TPVB.40 Multiple-injection TPVB produces more reliable radiographic spread and clinical distribution of anesthesia compared to a single-injection TPVB.46Paravertebral injections performed at 2-week intervals in the same patient also produce different degrees of sensory blockade.40 Thoracic paravertebral anesthesia does not appear to be gravity dependent, but there is a tendency for preferential caudal spread of somatic2,21,46 and sympathetic nerve blockade.2
USG TPVB also produces ipsilateral segmental thoracic anesthesia.11,44,47,48 The segmental spread of sensory blockade after a single-injection USG TPVB, using a transverse scan with out-of-plane needle insertion (lateral injection), is also reported as being variable and unpredictable.47 Data from more recent cadaver anatomic42 and clinical43-45 studies indicate that a USG TPVB may produce a different block dynamic than an anatomical landmark-based TPVB.42-45 The dermatomal distribution of anesthesia after an USG TPVB (paramedian sagittal in-plane approach) doesn’t appear to be influenced by the number of injections44 but is affected by the volume of injection (20 ml > 10 ml)42 and the position of the needle tip (LA injection) within the TPVS (medial approach > lateral approach).42,43 Epidural spread,42 postoperative analgesia,10 and reduction in blood pressure45 are also more significant when the paravertebral injection is performed close to the central neuraxis (medial approach > lateral approach).
Epidural Spread
There is controversy about the epidural spread and its role in TPVB extension. Postoperative infusion of radio-opaque contrast medium through an extrapleural paravertebral catheter remains confined to the paravertebral space.49However, 70% of percutaneous paravertebral injections show varying degrees of unilateral epidural spread,1, 35 but the volume involved is considered too small to produce a clinically significant epidural block.22 Cadaveric dissection also confirms that only a small proportion of the injectate enters the epidural space50 and remains confined to the side of the injection. The vertebral attachment of the endothoracic fascia limits prevertebral spread51 and may also influence epidural spread or mass movement of drugs after an extrapleural paravertebral compartment injection. Clinically, sensory anesthesia is predominantly ipsilateral and greater with epidural spread.35 Current evidence suggests that ipsilateral epidural spread of small amounts of LA contributes to the extension of a TPVB.
Contralateral Anesthesia
Segmental contralateral anesthesia occurs in 1.1% of paravertebral injections22 likely due to prevertebral34,52 or epidural35 spread to the contralateral TPVS. We have observed mean segmental contralateral anaesthesia of 2.5 (1.5) dermatomes in 20% of patients after a single-injection TPVB.41 More recently, we have reported contralateral anaesthesia of over 9 (7–11) dermatomes in 35% of patients after a multiple-injection TPVB that was used for surgical anaesthesia during percutaneous radiofrequency ablation of liver tumors.53 Bilateral symmetrical anesthesia is also described.35,41 It may be due to extensive epidural spread,35 inadvertent intrathecal injection into the dural sleeve,1 or inadvertent epidural injection via a catheter intended for the paravertebral space.41 Bilateral symmetrical anesthesia may also occur more commonly with the medial injection technique or large volumes of injectate (over 25 ml).1
Prevertebral Spread
The exact mechanism for prevertebral spread is not clear, but we have proposed that it occurs through the extrapleural compartment of the TPVS via the subserous layer of connective tissue.34 It is not known if there are differences in the incidence of contralateral anaesthesia between a single-injection and a multiple-injection TPVB, but our experience suggests that it may be more common after a multiple-injection technique.53 Bilateral sympathetic blockade may also occur in the absence of bilateral sensory blockade due to prevertebral spread34 to the contralateral sympathetic chain, which is located more anteriorly in the TPVS and is also more susceptible to LA. This may be one explanation for bilateral Horner’s syndrome reported after unilateral TPVB.35
Thoracolumbar Anesthesia
Ipsilateral lumbar spinal nerves are occasionally involved after a lower thoracic paravertebral injection.2,5 We have also observed ipsilateral radiological spread of contrast below the diaphragm.54 Saito and colleagues have demonstrated in volunteers that a single-injection TPVB at T11 with 22 ml of LA produces mean loss of sensation to pin-prick of 6 (8-13) segments, extending from T5 to L5 segments.55 Saito and colleagues have also demonstrated the ipsilateral thoracolumbar spread of colored dye in cadavers.56 These observations challenge the concept of lumbar nerve root sparing following TPVB.21
The Retro-SCTL Space and Its Role in Thoracic Anesthesia and Analgesia
Given the unique anatomy of the retro-SCTL space and its communications with the adjoining paraspinal fascial planes, TPVS, and neuraxis (described above),36,37 we believe it is an important crossroad for LA spread for the intertransverse process blocks (ITPB) or injections behind the ITTC.29,57 The presence of the ventral and dorsal rami of the thoracic spinal nerve, and the dorsal root ganglion within the medial aspect of the retro-SCTL space (Figures 5 and 6),36,37 also indicate that it may be possible to produce anesthesia and or analgesia of the ipsilateral hemithorax by injecting the LA behind the SCTL or ITTC.29,30 This is the proposed anatomic basis for an ITPB29,57 that has recently been described as an effective and potentially safer alternative to avoid pleural puncture30,31,58 than a TPVB. A detailed description of how an ITPB may affect the ventral and dorsal rami of the thoracic spinal nerve to produce anesthesia and/or analgesia of the ipsilateral hemithorax is beyond the scope of this article and discussed elsewhere.29
How I Perform Ultrasound-Guided Thoracic Paravertebral Block
Basic Considerations
An ultrasound scan for TPVB can be performed in the transverse or sagittal axis with the patient in the sitting, lateral decubitus, or prone position. The prone position is preferred in patients undergoing USG TPVB for chronic pain procedures because it allows the concurrent use of fluoroscopy. Since we routinely administer sedation (midazolam, dexmedetomidine) and analgesia (ketamine) for comfort during a TPVB, we position the patient in the lateral decubitus position with the side to be blocked uppermost (Figures 7 and 8). For consistency, we recommend using ultrasound to identify the thoracic vertebral level for the TPVB. No data demonstrates an optimal axis for the ultrasound scan or the injection. It is often a matter of individual choice or the experience of the proceduralist. The transducer of choice for the ultrasound scan also depends on the patient's body habitus. A literature review indicates high-frequency ultrasound (13-6 MHz) is more frequently used for USG TPVB9,59 than low-frequency ultrasound.1,12,60 This may be because the relevant anatomical structures (TP, SCTL, ITTC, and pleura) are relatively superficial in location, and high-frequency ultrasound, despite its limited penetration, provides higher resolution of these structures than a low-frequency ultrasound. However, in patients with large body habitus or when one must scan at a depth, a low-frequency ultrasound transducer (2-5 MHz) with a divergent beam and a wide field of view may be preferable.
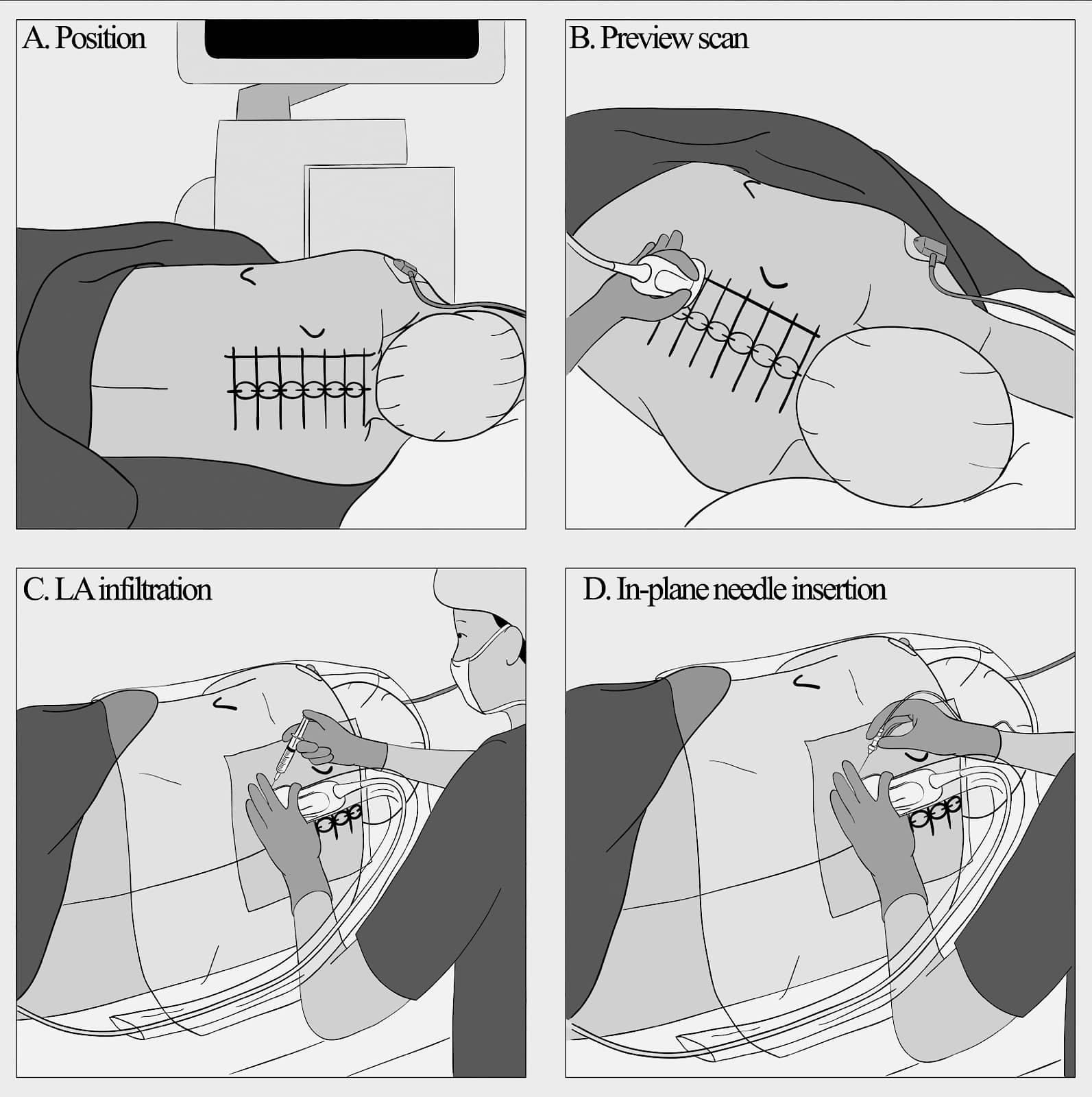
Figure 7. A series of illustrations showing patient position (A), position and orientation of the curvilinear transducer (B), local anesthetic infiltration (C), and in-plane needle insertion (D) during an ultrasound-guided thoracic paravertebral block.
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
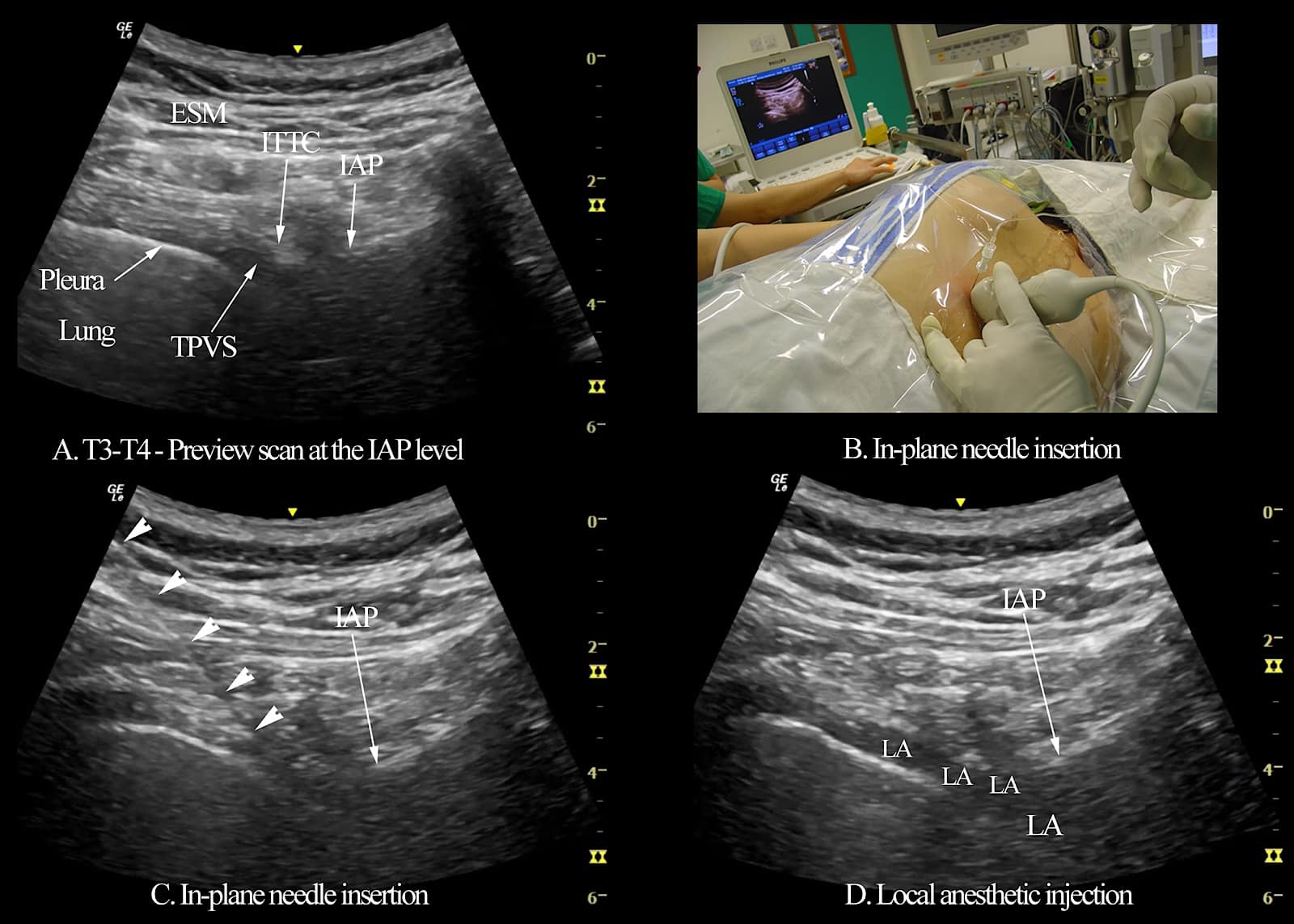
Figure 8. A series of images illustrating ultrasound-guided thoracic paravertebral block (TPVB) using a transverse scan at the inferior articular process (IAP) level, including the target transverse sonogram at the IAP level (A), the position of the patient and orientation of the curvilinear ultrasound transducer (B), in-plane needle insertion with white arrowheads showing the path of the block needle (C), and widening of the paravertebral space and spread of the local anesthetic to the posterior intercostal space (D).
TPVS = thoracic paravertebral space, ITTC = intertransverse tissue complex, ESM = the erector spinae muscle
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
Likewise, several different techniques have been described for performing USG TPVB.12 There are pros and cons of each technique, but there is a paucity of data comparing the safety and efficacy of the various approaches. Therefore, the choice of technique (as described above) also depends on the individual preference and experience of the anesthesiologist. Currently, the published literature indicates that USG TPVB is most frequently performed using a high-frequency linear transducer (13-6 MHz).9,61 To the best of our knowledge, there is a paucity of data on the use of low-frequency ultrasound for TPVB.11,12,60 However, contrary to common understanding (poor resolution), we have been using low-frequency ultrasound (2-5 MHz) to perform USG TPVB (Figures 7 and 8) for over a decade with great success and minimal complications (details below).11,62,63 A detailed description of the various USG techniques for TPVB is beyond the scope of this article and has been described elsewhere.12 The following section describes how we perform USG TPVB using a low-frequency curvilinear transducer.11,62,63
Rationale Behind the Transverse In-Plane Technique at the IAP Level
We use a low-frequency (2-5 MHz) curved array transducer to perform TPVB.11,62,63 A transverse scan of the thoracic paravertebral region is performed at the level of the IAP of the thoracic vertebra and the block needle is inserted in-plane from a lateral to the medial direction (Figures 7 and 8) aiming to place it at the apex of the TPVS (Figure 8).11,62,63 Our technique is a simple modification of the transverse ultrasound scan technique initially described by Shibata and Nishiwaki, who used high-frequency ultrasound to perform a transverse scan of the paravertebral region at the TP level.9 We modified this approach to maximize ultrasound visibility of the TPVS and reduce bony obstruction to the advancing block needle without the TP in the target ultrasound window (Figure 8).11,62,63 Our rationale behind using the transverse ultrasound scan at the IAP level with a low-frequency ultrasound (2-5 MHz) as the target window for the TPVB is as follows:
- There is minimal acoustic shadowing from bone, resulting in excellent ultrasound images of the paravertebral region (Figure 9)11,62,63
- The divergent ultrasound beam's wide field of view (Figures 8 and 9) offers an added advantage for visualizing the anatomy and guiding the block needle compared to the narrow rectangular field of view produced by a high-frequency linear array transducer.
- The ITTC and parietal pleura are clearly delineated in the transverse sonogram (Figures 8 and 9),11,62,63 but the pleura may not be as clearly delineated in a sagittal sonogram.
- The position of the intervertebral foramen can be easily determined in the transverse sonogram as it is located anterior to the IAP (Figure 9C), thereby establishing a “no-go zone” for needle placement.11,62,63
- There may be a lower potential for pleural puncture since the block needle is advanced tangential to the parietal pleura (Figure 8C).9
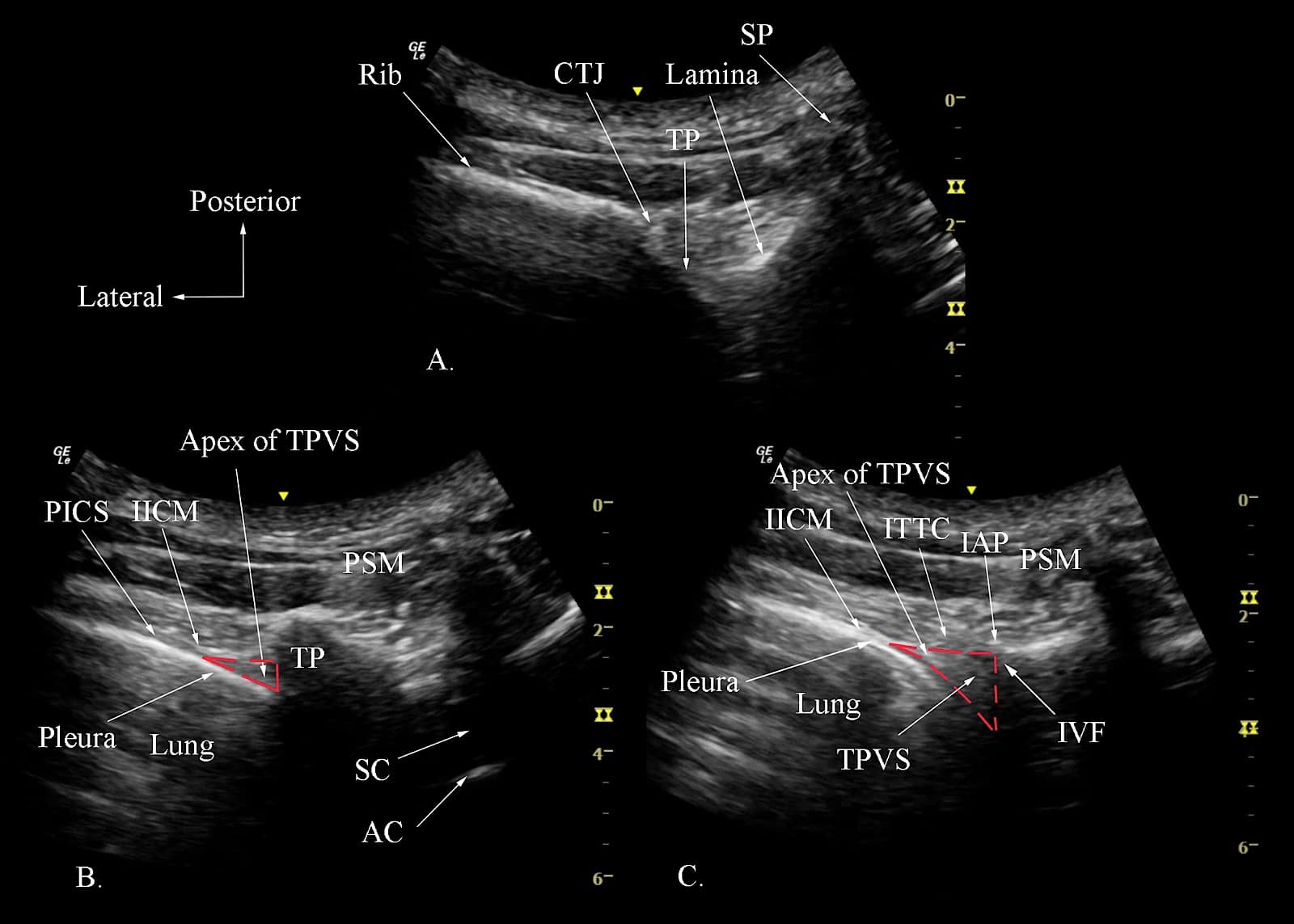
Figure 9. Transverse sonograms of the thoracic paravertebral region with the ultrasound beam being insonated at the level of the transverse process-rib complex (position 1 in Figure 10A) at the transverse process level, (position 2 in Figure 10B), and at the inferior articular process (IAP) level (position 3 in Figure 10C).
SP = spinous process, TP = transverse process, CTJ = costotransverse junction, PSM = paraspinal muscles, TPVS = thoracic paravertebral space, IICM = internal intercostal membrane, PICS = posterior intercostal space, SC = spinal canal, AC = anterior complex, ITTC = intertransverse tissue complex, IVF = intervertebral foramen
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
The Transverse Ultrasound Scan Sequence
The target transverse ultrasound window at the level of the IAP is obtained through three sequential steps, over three contiguous osseous anatomical landmarks (Figure 10), at the target vertebral level.11,62,63 The sequential steps are as follows: Step 1 at the transverse process-rib complex level (Figure 10, red box). Step 2 at the transverse process level (Figure 10, blue box), and step 3 at the IAP level (Figure 10, green box). Based on the underlying osseous and musculoskeletal structures, these three ultrasound scan windows produce three distinct sonograms (Figure 9). Correlative cadaver anatomic-CT-MRI and transverse sonograms for steps 2 and 3 of the transverse scan sequence are presented in Figures 11 and 12.
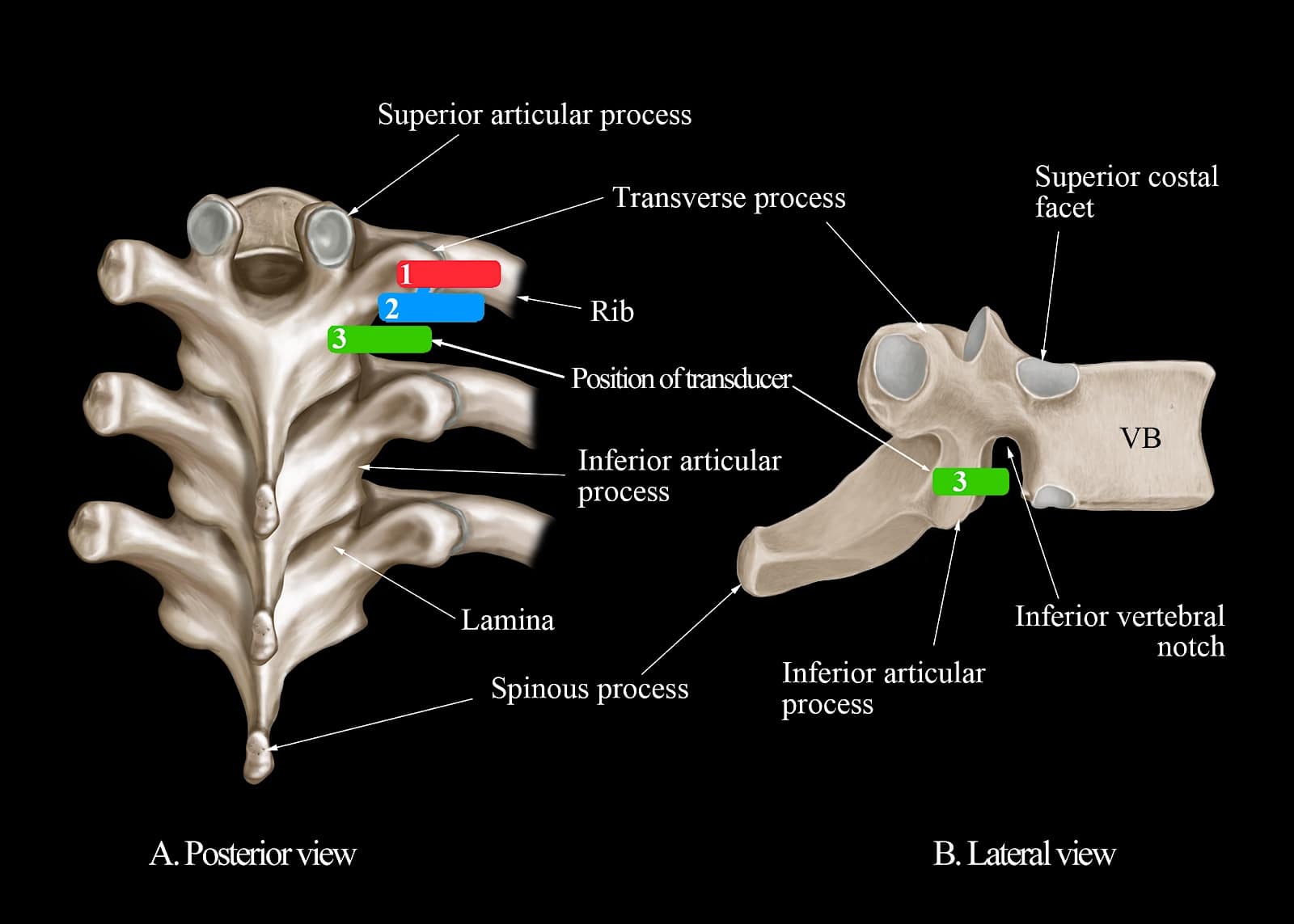
Figure 10. Illustrating the ultrasound transducer's position, relative to the underlying osseous anatomy, during the paramedian transverse ultrasound scan sequence described in this report. (A) Position 1 (red box) at the transverse process-rib complex level; position 2 (blue box) at the transverse process level; position 3 (green box) at the inferior articular process level. Note the anatomic relationship of the inferior articular process to the inferior vertebral notch and the intervertebral foramen. (B)
VB = vertebral body
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
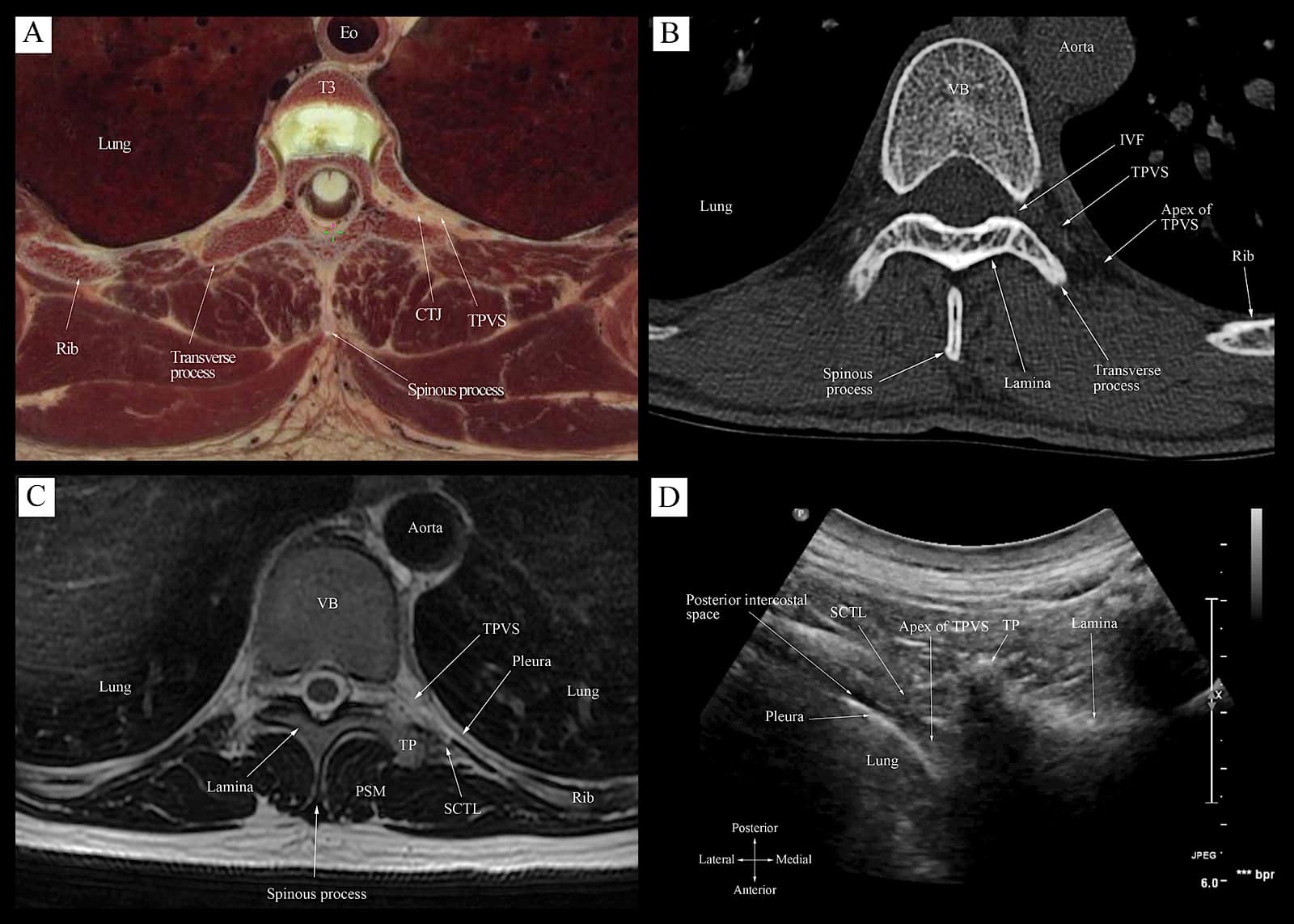
Figure 11. Correlative transverse cadaver anatomic (Fig 11A), CT (Fig 11B), MRI (T1 weighted, Fig 11C), and ultrasound (Fig 12D) images of the thoracic paravertebral region from the level of the vertebral body and transverse process corresponding to the level at which the transverse scan is performed (position 2, Fig 10).
Eo = esophagus, CTJ = costotransverse junction, TPVS = thoracic paravertebral space, VB = vertebral body, PSM = paraspinal muscle, IVF= intervertebral foramen, TP = transverse process, SCTL = superior costotransverse ligament
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
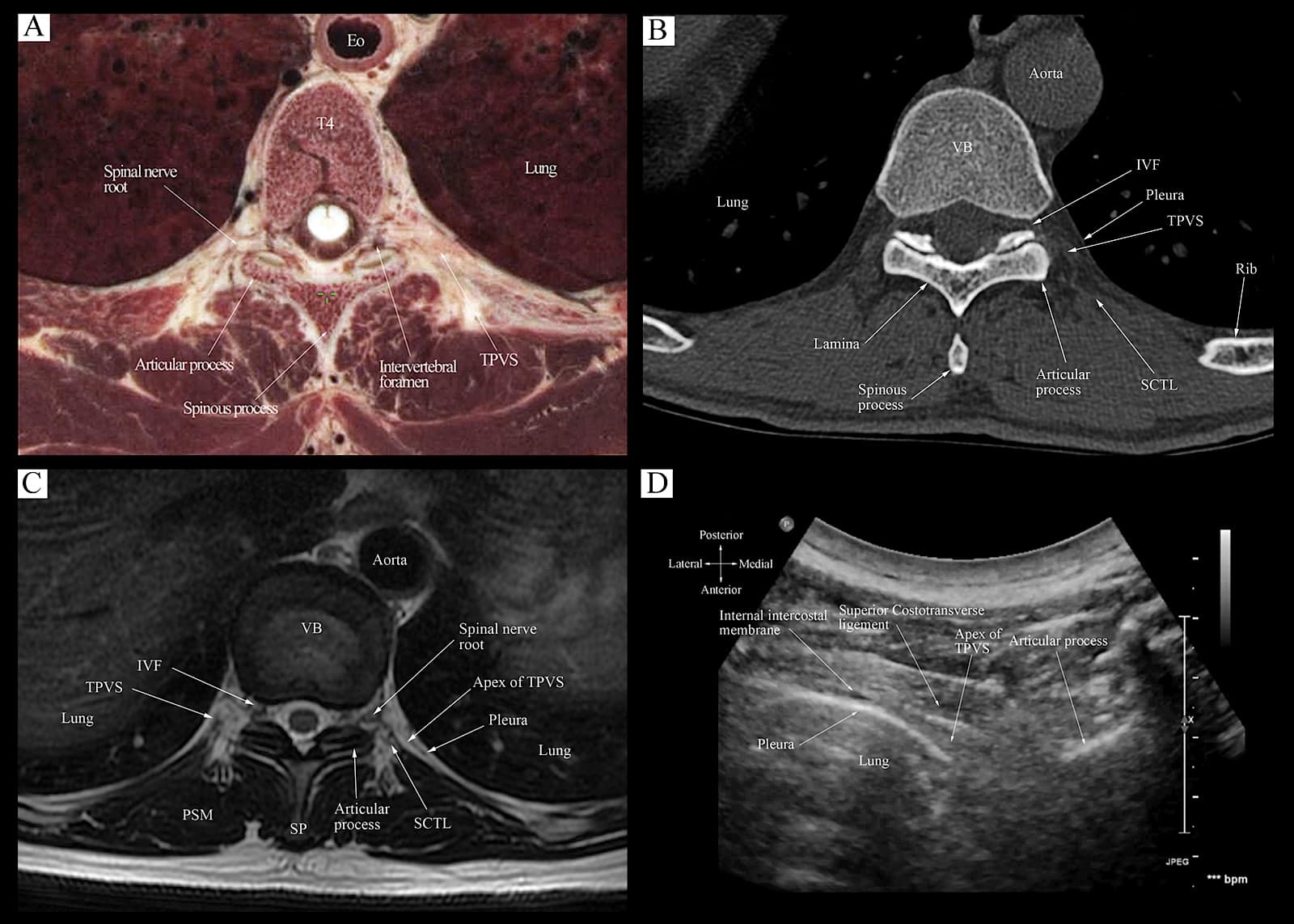
Figure 12. Correlative transverse cadaver anatomic (Fig 12A), CT (Fig 12B), MRI (T1 weighted, Fig 12C), and ultrasound (Fig 12D) images of the thoracic paravertebral region from the level of the vertebral body and inferior articular process corresponding to the level at which the transverse scan is performed (position 3, Fig 10).
TPVS = thoracic paravertebral space, VB = vertebral body, PSM = paraspinal muscle, IVF = intervertebral foramen, SCTL = superior costotransverse ligament, SP = spinous process
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
Step 1: Transverse scan at the transverse process-rib complex level. The transducer is positioned 2-3 cm lateral to the midline (spinous process) at the target vertebral level, in the transverse plane, and over the rib and costotransverse articulation (Figure 10, position 1, red box). The hyperechogenic outlines of the spinous process, lamina, TP, costo-transverse junction and the rib with their corresponding acoustic shadows are clearly delineated from a medial to lateral direction (Figure 9A). The posterior angulation of the TP of the thoracic vertebra is also easily recognized (Figure 9A). This contrasts with the TP of the lumbar vertebra, which is at right angles to the vertebral body. This ultrasound window does not lend itself to visualizing the paravertebral anatomy because the acoustic shadow of the TP-rib complex completely obscures the TPVS (Figure 9A). Therefore, it is used more as a starting point for the subsequent steps in the scan sequence.
Step 2: Transverse scan at the transverse process level. From position 1, gently slide the transducer caudally until the acoustic shadow of the rib is no longer visible (Figure 10, position 2, blue box), and the hyperechogenic outline of the lamina and TP with their acoustic shadow are visualized (Figure 9B).11,62,63 Lateral to the TP, one can define the IICM posteriorly and the hyperechogenic pleura and lung anteriorly. Interposed between the two is the hypoechoic apical part of the TPVS (Figure 9B).9,11,12 The PICS may also be visualized in continuity with the apex of the TPVS laterally (Figure 9B).
Step 3: Transverse scan at the inferior articular process level. Finally, suppose one gently slides or tilts the transducer caudally from position 2 (Figure 10, position 3, green box). In that case, the acoustic shadow of the lamina and transverse process disappear and the hyperechogenic IAP (Figure 9C) is now visualized medially.11,62,63 This acoustic window represents the transverse-intertransverse view of the TPVS.11,62,63 As in the sonogram at the level of the TP (Position 2, Figure 9B), the SCTL, parietal pleura, lung and the apical part of the paravertebral space are clearly delineated (Figure 9C).11,62,63 However, the area of the acoustic shadow at the level of the IAP is significantly less than that at the level of the TP. As a result, one can visualize the majority of the TPVS in this view (Figure 9C).11,62,63 On a different note, the same transverse ultrasound window at the IAP level can be utilized to visualize the retro-SCTL space by gently pivoting the lateral end of the transducer caudally and performing an ITPB at the retro-SCTL space.29,57 Currently, limited published data are describing the use of a transverse scan at the level of the IAP for USG TPVB.11,12,63
USG TPVB Using the Transverse In-plane Approach at the IAP Level
Under strict aseptic precautions, and with the target transverse ultrasound window at the IAP level in view (Figure 8), the nerve block needle is introduced in-plane from a lateral-to-medial direction (Figure 8B).11,62,63 The needle is advanced under real-time ultrasound guidance with a gentle back-and-forth (jiggling) motion until the needle tip is embedded within the ITTC. Interestingly, it is not uncommon to lose track of the needle tip once it enters the ITTC, possibly because the needle and ITTC are isoechoeic (Figure 8C). The needle is advanced in small increments followed by a 1-2 ml of 0.9% saline test bolus. Swelling of the ITTC or an anechoic fluid-filled area within the ITTC indicates that the needle tip is still within the ITTC. Entry of the needle tip into the TPVS is visualized as anterior displacement of the parietal pleura, widening of the paravertebral space, and increased pleural echogenicity upon saline injection (Figures 8D and 13).11,62,63 A calculated dose of LA is then injected in small aliquots.11,62-64 For multi-level USG TPVB, such as for primary breast cancer surgery, we use 3-4 ml of 0.5% ropivacaine or levobupivacaine with 1:200,000 epinephrine per level, with the total volume capped at 25 ml (125 mg) for the 6 injections (T1-T6).11,62,63 For perioperative analgesia, 20-25 ml of the same LA is injected at a single-level (T3-T4 for PBCS).64
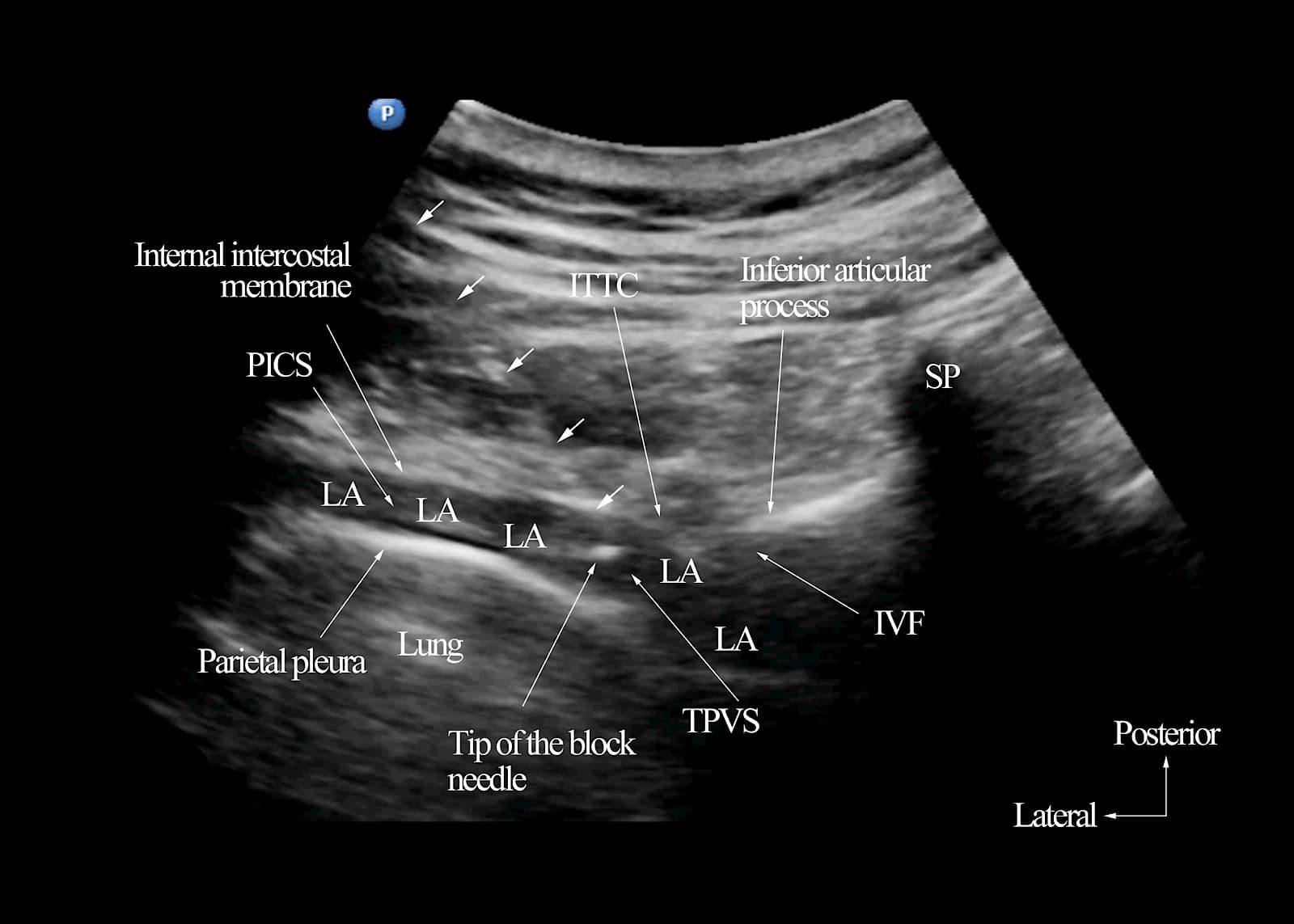
Figure 13. Transverse sonogram of the thoracic paravertebral region after a single-injection ultrasound-guided thoracic paravertebral block (25 ml volume) at the inferior articular process level. Note the position of the intervertebral foramen (IVF) relative to the inferior articular process. Multiple small white arrows have been used to demonstrate the path of the block needle. The tip of the block needle is near the apex of the paravertebral space and away from the IVF. Also, note there is a widening of the thoracic paravertebral space (TPVS) and spread of the local anesthetic to the medial aspect of the TPVS and laterally to the posterior intercostal space.
SP = spinous process, ITTC = intertransverse tissue complex
Reproduced with permission from the Department of Anesthesia and Intensive Care, The Chinese University of Hong Kong, Faculty of Medicine, www.aic.cuhk.edu.hk/usgraweb.
Indications
TPVB is indicated for anesthesia and analgesia when the nociceptive input is predominantly unilateral in origin from the thorax and abdomen. Some of the commonly reported indications for a TPVB are listed in Table 1.1
A. Surgical Anaesthesia
|
B. Perioperative Analgesia
|
C. Acute and Chronic Pain Management
|
D. Miscellaneous
|
Complications
Major complications after a TPVB are rare.1,22 However, there is a paucity of data on complications specifically associated with USG TPVB,62,65-67 and no comparative studies have evaluated complication rates between anatomical landmark-based TPVB and USG TPVB. Pleural puncture and postoperative pneumothorax are uncommon. Niesen et al.66 reported a 0.38% incidence of postoperative pneumothorax, while Kelly et al. documented pleural puncture and pneumothorax rates of 0.6% and 0.26%, respectively.67 Using the transverse in-plane approach at the IAP level (described above), we report a zero incidence of pneumothorax following USG TPVB.62 These rates agree with the 0.5% incidence associated with landmark-based TPVB techniques.1,22 However, the zero incidence of pleural puncture should be interpreted cautiously as these figures come from retrospective studies that may be biased. Additional complications after USG TPVB include vascular puncture, hypotension (particularly using the transverse technique described),1,22,45,62 Horner’s syndrome,1,41,62 recurrent laryngeal nerve palsy,62 and local anesthetic systemic toxicity.1,68
Conclusion
Thoracic paravertebral block (TPVB) reliably produces ipsilateral somatic and sympathetic nerve blockade across multiple thoracic dermatomes. USG TPVB significantly advances conventional landmark-based techniques by offering improved precision and safety. Modern ultrasound technology allows detailed visualization of relevant anatomic structures, facilitating accurate needle placement and minimizing the risk of pleural puncture. Real-time ultrasound guidance also enables clinicians to observe LA dispersion during administration, enhancing block efficacy and predictability. This report describes our preferred transverse in-plane technique at the inferior articular process level for USG TPVB, which we have been using successfully for over a decade. Currently, data comparing various USG TPVB techniques are limited. Future research to establish the optimal technique for clinical practice is warranted.


References
- Karmakar M. Thoracic paravertebral block. Anesthesiology 2001;95:771-80. https://doi.org/10.1097/00000542-200109000-00033
- Cheema SPS, Ilsley D, Richardson J, et al. A thermographic study of paravertebral analgesia.Anaesthesia 1995;50:118-21. https://doi.org/10.1111/j.1365-2044.1995.tb15092.x
- Lund C, Selmar P, Hansen OB, et al. Effect of epidural bupivacaine on somatosensory evoked potentials after dermatomal stimulation. Anesth Analg 1987;66:34-8.
- Lund C, Selmar P, Hansen OB, et al. Effect of intrathecal bupivacaine on somatosensory evoked potentials following dermatomal stimulation. Anesth Analg 1987;66:809-13.
- Richardson J, Jones J, Atkinson R. The effect of thoracic paravertebral blockade on intercostal somatosensory evoked potentials. Anesthesia and Analgesia 1998;87:373-6. https://doi.org/10.1097/00000539-199808000-00025
- Greengrass R, O'Brien F, Lyerly K, et al. Paravertebral block for breast cancer surgery. Can J Anaesth1996;43:858-61. https://doi.org/10.1007/BF03013039
- Klein SM, Greengrass RA, Weltz C, et al. Paravertebral somatic nerve block for outpatient inguinal herniorrhaphy: an expanded case report of 22 patients. Reg Anesth Pain Med 1998;23:306-10. https://doi.org/10.1016/s1098-7339(98)90059-1
- Richardson J, Vowden P, Sabanathan S. Bilateral paravertebral analgesia for major abdominal vascular-surgery - a preliminary-report. Anaesthesia 1995;50:995-8. https://doi.org/10.1111/j.1365-2044.1995.tb05939.x
- Shibata Y, Nishiwaki K. Ultrasound-guided intercostal approach to thoracic paravertebral block. Anesth Analg 2009;109:996-7. https://doi.org/10.1213/ane.0b013e3181af7e7b
- Taketa Y, Fujitani T, Irisawa Y, et al. Ultrasound-guided thoracic paravertebral block by the paralaminar in-plane approach using a microconvex array transducer: methodological utility based on anatomical structures.J Anesth 2017;31:271-7. https://doi.org/10.1007/s00540-016-2289-8
- Pangthipampai P, Karmakar MK, Songthamwat B, et al. Ultrasound-guided multilevel thoracic paravertebral block and its efficacy for surgical anesthesia during primary breast cancer surgery. J Pain Res2020;13:1713-23. https://doi.org/10.2147/JPR.S246406
- Krediet AC, Moayeri N, van Geffen GJ, et al. Different approaches to ultrasound-guided thoracic paravertebral block: An illustrated review. Anesthesiology 2015;123:459-74. https://doi.org/10.1097/ALN.0000000000000747
- Richardson J, Lonnqvist PA. Thoracic paravertebral block. Br J Anaesth 1998;81:230-8. https://doi.org/10.1093/bja/81.2.230
- Richardson J, Sabanathan S. Thoracic paravertebral analgesia. Acta Anaesthesiol Scand 1995;39:1005-15. https://doi.org/10.1111/j.1399-6576.1995.tb04219.x
- Eason MJ, Wyatt R. Paravertebral thoracic block - reappraisal. Anaesthesia 1979;34:638-42. https://doi.org/10.1111/j.1365-2044.1979.tb06363.x
- Lönnqvist PA. Continuous paravertebral block in children. Initial experience. Anaesthesia 1992;47:607-9. https://doi.org/10.1111/j.1365-2044.1992.tb02336.x
- Karmakar MK, Booker PD, Franks R, et al. Continuous extrapleural paravertebral infusion of bupivacaine for post-thoracotomy analgesia in young infants. Br J Anaesth 1996;76:811-5. https://doi.org/10.1093/bja/76.6.811
- Eng J, Sabanathan S. Continuous paravertebral block for postthoracotomy analgesia in children. J Pediatr Surg 1992;27:556-7. https://doi.org/10.1016/0022-3468(92)90444-c
- Richardson J, Sabanathan S, Jones J, et al. A prospective, randomized comparison of preoperative and continuous balanced epidural or paravertebral bupivacaine on post-thoracotomy pain, pulmonary function and stress responses. Br J Anaesth 1999;83:387-92. https://doi.org/10.1093/bja/83.3.387
- Richardson J, Sabanathan S, Mearns AJ, et al. A prospective, randomized comparison of interpleural and paravertebral analgesia in thoracic-surgery. Br J Anaesth 1995;75:405-8. https://doi.org/10.1093/bja/83.3.387
- Lonnqvist PA, Hildingsson U. The caudal boundary of the thoracic paravertebral space - a study in human cadavers. Anaesthesia 1992;47:1051-2. https://doi.org/10.1111/j.1365-2044.1992.tb04200.x
- Lonnqvist PA, Mackenzie J, Soni AK, Conacher ID. Paravertebral blockade - failure rate and complications. Anaesthesia 1995;50:813-5. https://doi.org/10.1111/j.1365-2044.1995.tb06148.x
- Lönnqvist PA, Richardson J. Use of paravertebral blockade in children. Techniques in Regional Anesthesia & Pain Management 1999;3:184-8.
- Richardson J, Lonnqvist PA, Naja Z. Bilateral thoracic paravertebral block: Potential and practice. Br J Anaesth 2011;106:164-71. https://doi.org/10.1093/bja/aeq378
- Kapral S, Krafft P, Eibenberger K, et al. Ultrasound-guided supraclavicular approach for regional anesthesia of the brachial plexus. Anesth Analg 1994;78:507-13. https://doi.org/10.1213/00000539-199403000-00016
- Hara K, Sakura S, Nomura T. [Use of ultrasound for thoracic paravertebral block]. Masui 2007;56:925-31.
- Luyet C, Eichenberger U, Greif R, et al. Ultrasound-guided paravertebral puncture and placement of catheters in human cadavers: an imaging study. Br J Anaesth 2009;102:534-9. https://doi.org/10.1093/bja/aep015
- Yang HM, Choi YJ, Kwon HJ, et al. Comparison of injectate spread and nerve involvement between retrolaminar and erector spinae plane blocks in the thoracic region: a cadaveric study. Anaesthesia2018;73:1244-50. https://doi.org/10.1111/anae.14408
- Karmakar MK, Sivakumar RK, Sheah K, et al. Quest for the elusive mechanism of action for the thoracic paraspinal nerve block techniques. Are we ignoring the anatomy of the "retro superior costotransverse ligament space?".Anesth Analg 2023;137:458-65. https://doi.org/10.1213/ANE.0000000000006462
- Costache I, de Neumann L, Ramnanan CJ, et al. The mid-point transverse process to pleura (MTP) block: A new end-point for thoracic paravertebral block. Anaesthesia 2017;72:1230-6. https://doi.org/10.1111/anae.14004
- Nielsen MV, Moriggl B, Hoermann R, et al. Are single-injection erector spinae plane block and multiple-injection costotransverse block equivalent to thoracic paravertebral block? Acta Anaesthesiol Scand2019;63:1231-8. https://doi.org/10.1111/aas.13424
- Nielsen MV, Moriggl B, Borglum J. The intertransverse tissue complex: friend, foe, fallacies? Reg Anesth Pain Med 2021;46:379. https://doi.org/10.1136/rapm-2020-101583
- Karmakar M, Chung D. Variability of a thoracic paravertebral block. Are we ignoring the endothoracic fascia? Reg Anesth Pain Med 2000;25:325-7. https://doi.org/10.1016/s1098-7339(00)90028-2
- Karmakar MK, Kwok WH, Kew J. Thoracic paravertebral block: radiological evidence of contralateral spread anterior to the vertebral bodies. Br J Anaesth 2000;84:263-5. https://doi.org/10.1093/oxfordjournals.bja.a013417
- Purcell-Jones G, Pither CE, Justins DM. Paravertebral somatic nerve block: a clinical, radiographic, and computed tomographic study in chronic pain patients. Anesth Analg 1989;68:32-9.
- Cho TH, Kim SH, O J, et al. Anatomy of the thoracic paravertebral space: 3d micro-ct findings and their clinical implications for nerve blockade. Reg Anesth Pain Med 2021;46:699-703. https://doi.org/10.1136/rapm-2021-102588
- Cho TH, Kwon HJ, O J, et al. The pathway of injectate spread during thoracic intertransverse process (ITP) block: Micro-computed tomography findings and anatomical evaluations. J Clin Anesth 2022;77:110646. https://doi.org/10.1016/j.jclinane.2022.110646
- Kim SH. Anatomical classification and clinical application of thoracic paraspinal blocks. Korean J Anesthesiol 2022;75:295-306. https://doi.org/10.4097/kja.22138
- Choi YJ, Kwon HJ, O J et al. Influence of injectate volume on paravertebral spread in erector spinae plane block: an endoscopic and anatomical evaluation. PLoS One 2019;14:e0224487. https://doi.org/10.1371/journal.pone.0224487
- Cheema S, Richardson J, McGurgan P. Factors affecting the spread of bupivacaine in the adult TPV space. Anaesthesia 2003;58:684-711. https://doi.org/10.1046/j.1365-2044.2003.03189_1.x
- Karmakar M, Critchley L, Ho A, et al. Continuous thoracic paravertebral infusion of bupivacaine for pain management in patients with multiple fractured ribs. Chest 2003;123:424-31. https://doi.org/10.1378/chest.123.2.424
- Seidel R, Wree A, Schulze M. Thoracic-paravertebral blocks: comparative anatomical study with different injection techniques and volumes. Reg Anesth Pain Med 2020;45:102-6. https://doi.org/10.1136/rapm-2019-100896
- Taketa Y, Irisawa Y, Fujitani T. Comparison of analgesic efficacy between two approaches of paravertebral block for thoracotomy: a randomised trial. Acta Anaesthesiol Scand 2018;62:1274-9. https://doi.org/10.1111/aas.13216
- Uppal V, Sondekoppam RV, Sodhi P, et al. Single-injection versus multiple-injection technique of ultrasound-guided paravertebral blocks: a randomized controlled study comparing dermatomal spread. Reg Anesth Pain Med 2017;42:575-81. https://doi.org/10.1097/AAP.0000000000000631
- Wu ZW, Ni HD, Hou XM, et al. [effects of lateral and medial points of thoracic paravertebral nerve block by ultrasound for rapid recovery after laparoscopic cholecystectomy]. Zhonghua Yi Xue Za Zhi2019;99:988-92. https://doi.org/10.3760/cma.j.issn.0376-2491.2019.13.006
- Naja ZM, El-Rajab M, Al-Tannir MA et al. Thoracic paravertebral block: influence of the number of injections. Reg Anesth Pain Med 2006;31:196-201. https://doi.org10.1016/j.rapm.2005.12.004/
- Marhofer D, Marhofer P, Kettner SC et al. Magnetic resonance imaging analysis of the spread of local anesthetic solution after ultrasound-guided lateral thoracic paravertebral blockade. Anesthesiology 2013 May;118:1106-12. https://doi.org/10.1097/ALN.0b013e318289465f
- Patnaik R, Chhabra A, Subramaniam R et al. Comparison of paravertebral block by anatomic landmark technique to ultrasound-guided paravertebral block for breast surgery anesthesia: a randomized controlled trial.Reg Anesth Pain Med 2018;43:385-90. https://doi.org/10.1097/AAP.0000000000000746
- Eng J, Sabanathan S. Site of action of continuous extrapleural intercostal nerve block. Ann Thorac Surg1991;51:387-9. https://doi.org/10.1016/0003-4975(91)90850-p
- Conacher ID. Resin injection of thoracic paravertebral spaces. Br J Anaesth 1988;61:657-61. https://doi.org/10.1093/bja/61.6.657
- Moore DC, Bush WH, Scurlock JE. Intercostal nerve block: a roentgenographic anatomic study of technique and absorption in humans. Anesth Analg 1980;59:815-25.
- Karmakar M, Chui P, Joynt G, et al. Thoracic paravertebral block for management of pain associated with multiple fractured ribs in patients with concomitant lumbar spinal trauma. Reg Anesth Pain Med 2001;26:169-73. https://doi.org/10.1053/rapm.2001.21086
- Cheung N, Karmakar M. Right thoracic paravertebral anaesthesia for percutaneous radiofrequency ablation of liver tumours. Br J Radiol 2011;84:785-9. https://doi.org/10.1259/bjr/28983063
- Karmakar MK, Gin T, Ho AMH. Ipsilateral thoraco-lumbar anaesthesia and paravertebral spread after low thoracic paravertebral injection. Br J Anaesth 2001;87:312-6. https://doi.org/10.1093/bja/87.2.312
- Saito T, Den S, Cheema SP et al. A single-injection, multi-segmental paravertebral block-extension of somatosensory and sympathetic block in volunteers. Acta Anaesthesiol Scand 2001;45:30-3. https://doi.org/0.1034/j.1399-6576.2001.450105.x
- Saito T, Den S, Tanuma K, et al. Anatomical bases for paravertebral anesthetic block: fluid communication between the thoracic and lumbar paravertebral regions. Surg Radiol Anat 1999;21:359-63. https://doi.org/10.1007/BF01631341
- Karmakar MK, Sivakumar RK, Sheah K, et al. The retro superior costotransverse ligament space as a new target for ultrasound-guided intertransverse process block: a report of 2 cases. A A Pract 2022;16:e01610. https://doi.org/10.1213/XAA.0000000000001610
- Shibata Y, Kampitak W, Tansatit T. The novel costotransverse foramen block technique: distribution characteristics of injectate compared with erector spinae plane block. Pain Physician 2020;23:E305-14.
- Hara K, Sakura S, Nomura T, et al. Ultrasound-guided thoracic paravertebral block in breast surgery. Anaesthesia 2009;64:223-5. https://doi.org/10.1111/j.1365-2044.2008.05843.x
- Luyet C, Herrmann G, Ross S et al. Ultrasound-guided thoracic paravertebral puncture and placement of catheters in human cadavers: where do catheters go? Br J Anaesth 2011;106:246-54. https://doi.org/10.1093/bja/aeq309
- Marhofer P, Kettner SC, Hajbok L, et al. Lateral ultrasound-guided paravertebral blockade: an anatomical-based description of a new technique. Br J Anaesth 2010;105:526-32. https://doi.org/10.1093/bja/aeq206
- Karmakar MK, Pakpirom J, Sivakumar RK, et al. Ultrasound-guided thoracic paravertebral block using a transverse in-plane approach at the level of the inferior articular process: a retrospective review of complications. Reg Anesth Pain Med 2024;49(10):770-2. https://doi.org/10.1136/rapm-2022-103983
- Karmakar MK, Pakpirom J, Songthamwat B, et al. Subpectoral plexus block to enhance surgical anesthesia produced by a multilevel thoracic paravertebral block for primary breast cancer surgery: a prospective randomized double-blind study. Reg Anesth Pain Med 2025; rapm-2024-106126. https://doi.org/10.1136/rapm-2024-106126
- Karmakar MK, Samy W, Li JW, et al. Thoracic paravertebral block and its effects on chronic pain and health-related quality of life after modified radical mastectomy. Reg Anesth Pain Med 2014;39:289-98. https://doi.org/10.1097/AAP.0000000000000113
- Pace MM, Sharma B, Anderson-Dam J, et al. Ultrasound-guided thoracic paravertebral blockade: a retrospective study of the incidence of complications. Anesth Analg 2016;122:1186-91. https://doi.org/10.1213/ANE.0000000000001117
- Niesen AD, Jacob AK, Law LA, et al. Complication rate of ultrasound-guided paravertebral block for breast surgery. Reg Anesth Pain Med 2020;45:813-7. https://doi.org/10.1136/rapm-2020-101402
- Kelly ME, Mc Nicholas D, Killen J, et al. Thoracic paravertebral blockade in breast surgery: is pneumothorax an appreciable concern? a review of over 1000 cases. Breast J 2018;24:23-7. https://doi.org/10.1111/tbj.12831
- Fagenholz PJ, Bowler GM, Carnochan FM, et al. Systemic local anaesthetic toxicity from continuous thoracic paravertebral block. Br J Anaesth 2012;109:260-2. https://doi.org/10.1093/bja/aes126