How I Do It: Suprazygomatic Maxillary Nerve Block in Pediatrics
Cite as: Ali FM, Sunder A, Bolnick DA, et al. How I do it: suprazygomatic maxillary nerve block in pediatrics. ASRA Pain Medicine News 2026;51. https://doi.org/10.52211/asra050126.010.
How I Do It
Introduction
Maxillofacial surgery is frequently associated with significant postoperative pain and an increased risk of upper airway obstruction, including related respiratory complications. Opioid-based analgesia may further exacerbate these risks. The use of regional nerve blocks can enhance perioperative analgesia, promote hemodynamic stability, and reduce opioid-related adverse effects.1,2 Specifically, maxillary nerve blockade in the pterygopalatine fossa provides effective analgesia for select maxillofacial and upper airway procedures. Ultrasound guidance further improves the efficacy and safety of this technique by enabling visualization of key anatomical landmarks and confirmation of local anesthetic spread. Below, we review the relevant anatomy, sonoanatomy, technique, indications, and contraindications for performing a suprazygomatic maxillary (SZM) nerve block.
Anatomy
Maxillary Nerve
The maxillary nerve is the second division of the trigeminal nerve, also known as CN-V or cranial nerve five (Figure 1). This purely sensory nerve provides innervation to the mid-face, including the lower eyelid, the side of the nose, the upper lip and dental arch, the hard palate, soft palate, tonsils, nasal cavity, maxillary sinus, and the dura mater of the middle cranial fossa (MCF).
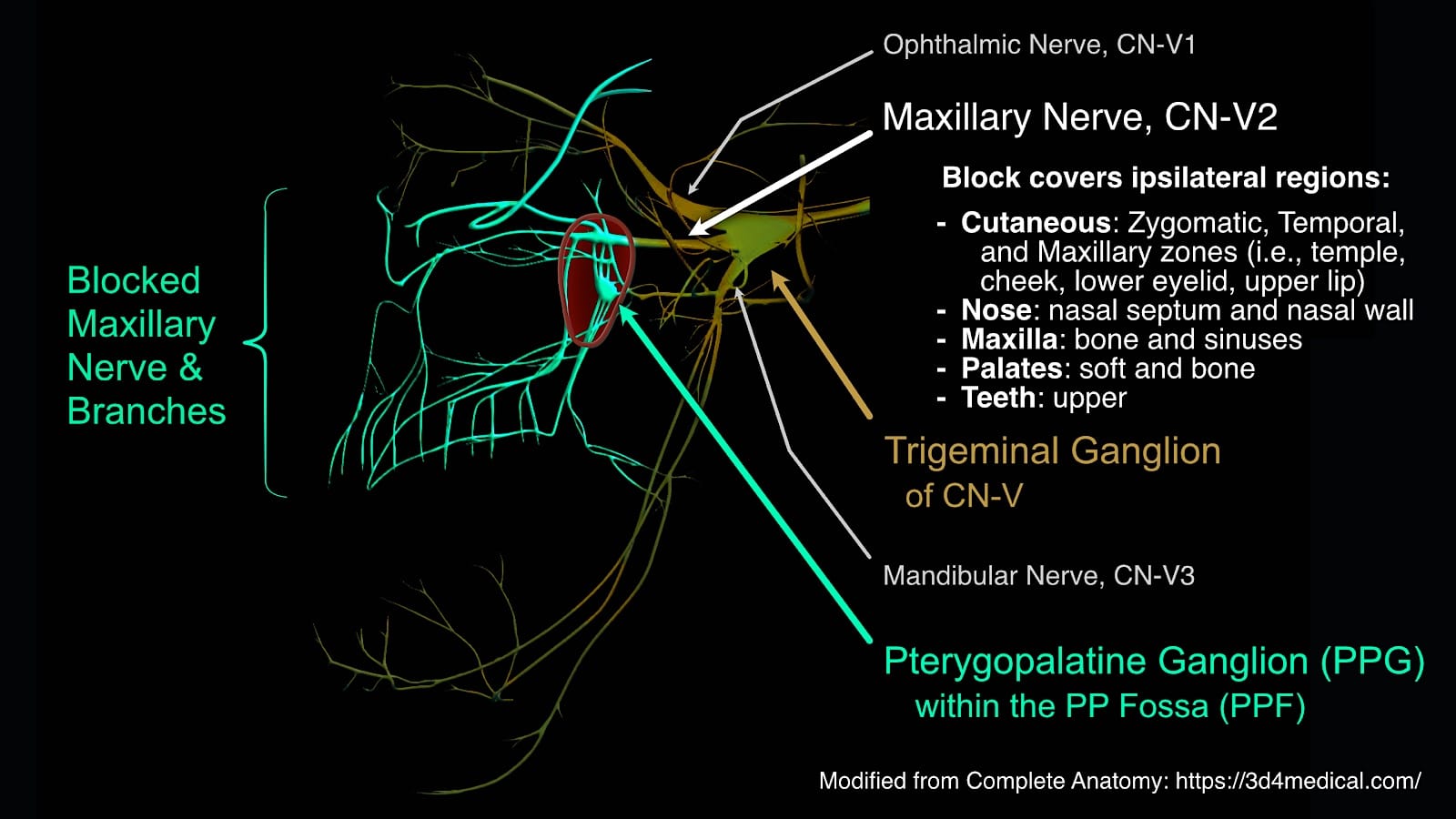
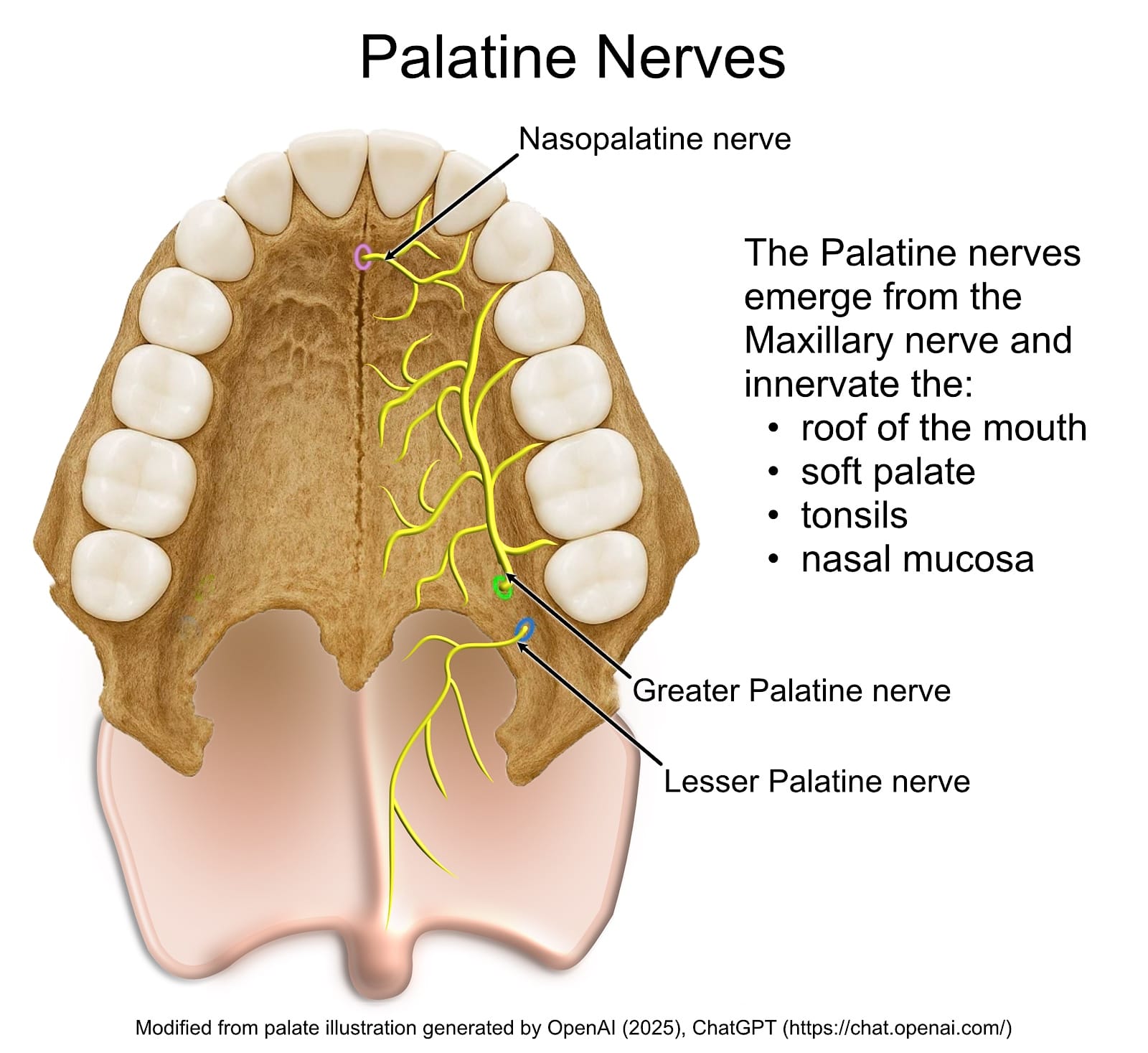
After emerging from the trigeminal ganglion, the maxillary nerve leaves the skull through the foramen rotundum and passes through the pterygopalatine fossa (PPF). It gives off several branches within the PPF before leaving the fossa through the inferior orbital fissure as the infraorbital nerve. The branches of the maxillary nerve in the PPF are the zygomatic nerve, the posterior superior alveolar nerve, and two ganglionic pterygopalatine branches to the pterygopalatine ganglion. The pterygopalatine ganglion then arborizes into the greater and lesser palatine nerves (Figure 2), nasopalatine nerve, posterior/lateral nasal branches nerves, and pharyngeal branch nerve.3 These branches together carry sensation from the maxillary teeth and sinus, nasal cavity, nasopharynx, and palate, explaining the broad coverage of a maxillary nerve block in the PPF.
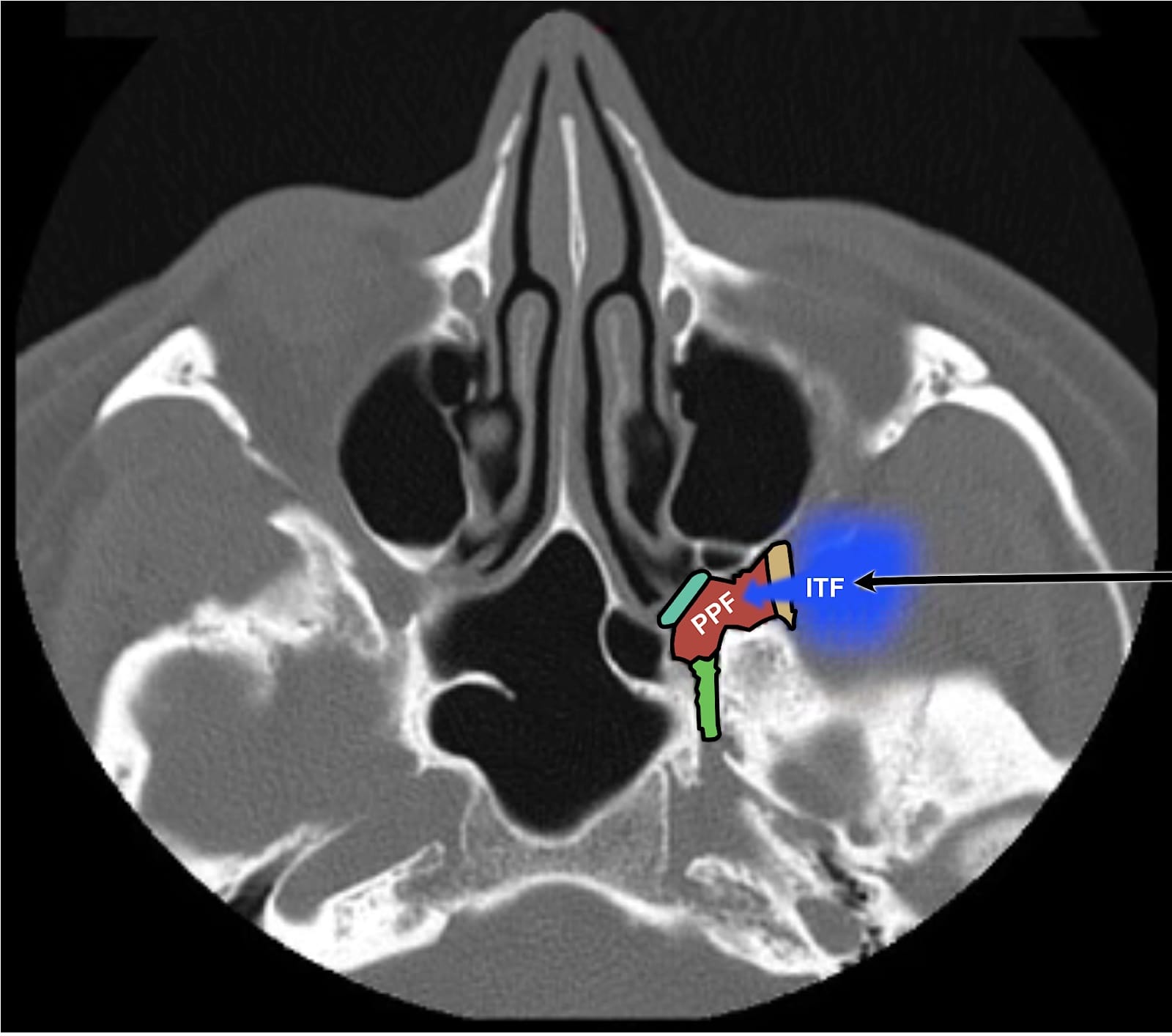
Pterygopalatine Fossa and Infratemporal Fossa
Understanding the anatomy of the PPF and the infratemporal fossa (ITF) is crucial for accurate and safe placement of local anesthetic during nerve blockade.
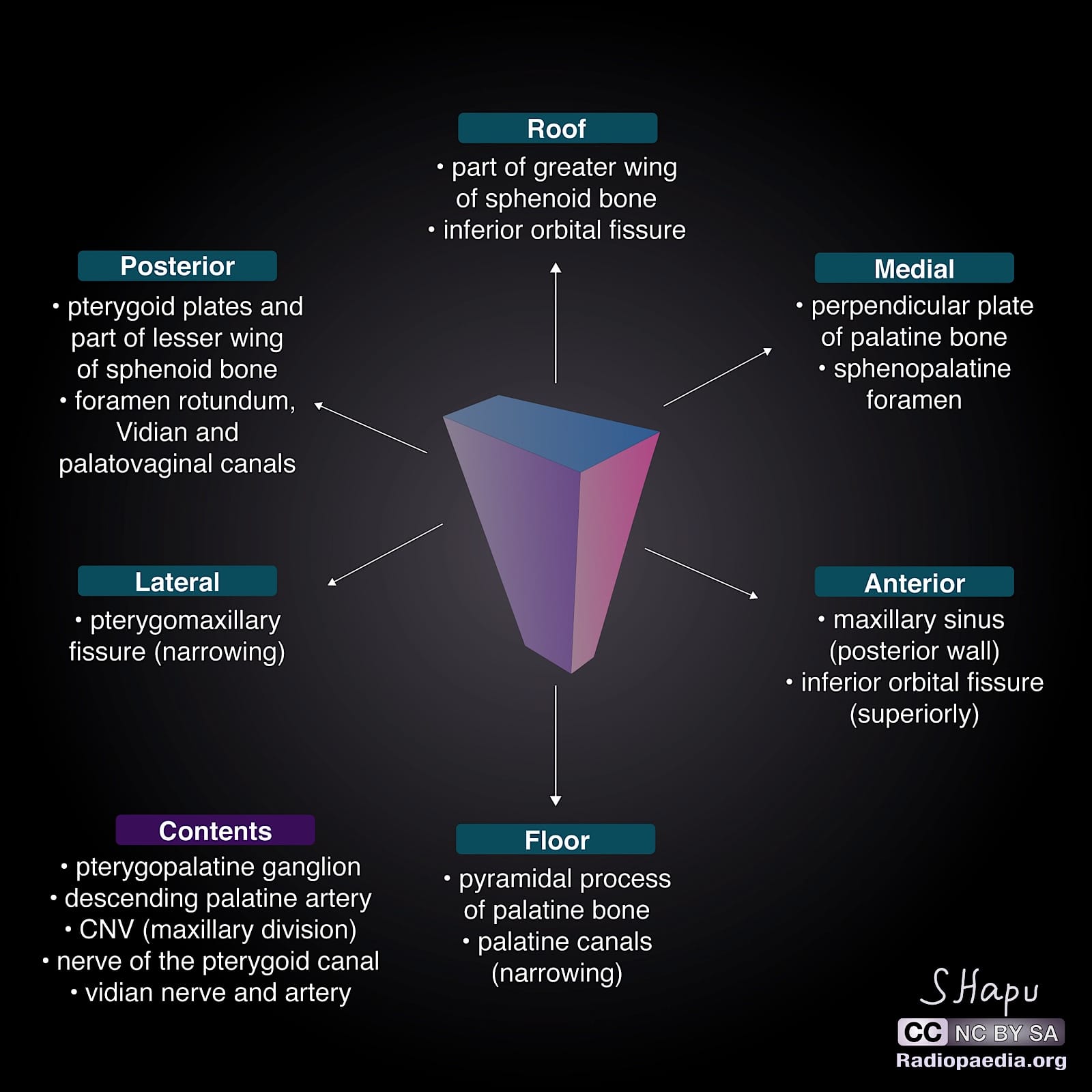
The PPF, also known as the sphenopalatine fossa, is a small, tapered cavity located deep in the midface, bounded by the maxilla as the anterior wall, the palatine as the medial wall and floor, and the sphenoid as the posterior wall and roof (Figure 3). It has an approximate volume of 1ml in adults,4 and possibly even smaller in children. It contains important neurovascular structures, including the maxillary division (CN-V2) of the trigeminal nerve and its branches, the pterygopalatine ganglion, distal branches of the maxillary artery, and emissary veins. The PPF communicates laterally with the more superficial infratemporal fossa through the narrow pterygomaxillary fissure. It also communicates with the orbit, nasal cavity, oral cavity, MCF, and pharynx through various foramina and canals (Figure 4).5
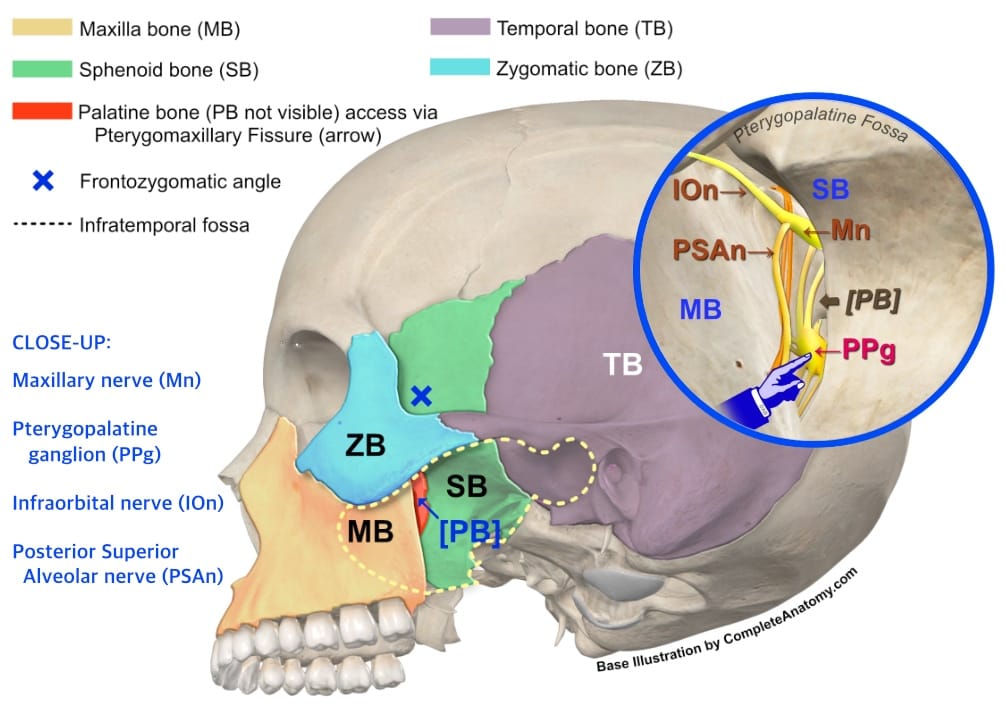
(Amini B, Schmidt R, Chieng R. Pterygopalatine fossa. Radiopaedia; 2009. https://doi.org/10.53347/rID-6969
The ITF6 is the anatomical space inferior to the zygomatic arch. Its boundaries are defined by the mandibular ramus (lateral), lateral pterygoid plate (medial), posterior maxilla (anterior), mastoid portion of the temporal bone (posterior), and the greater wing of the sphenoid (roof). The ITF connects to the temporal fossa, PPF, MCF, and orbit, acting as a crucial pathway for neurovascular structures. Key contents include the temporalis and pterygoid muscles, the maxillary artery, pterygoid venous plexus, retromandibular vein, and the mandibular nerve (CN-V3) and its branches.
Because the PPF is so small, a local anesthetic is injected into the infratemporal fossa, adjacent to the “mouth” of the PPF. The injectate then tracks through the pterygomaxillary fissure into the PPF to bathe the maxillary nerve and the pterygopalatine ganglion (Figure 5).4
The base image is from Amini B, Schmidt R, Chieng R. Pterygopalatine fossa. Radiopaedia; 2009. https://doi.org/10.53347/rID-6969
Approaches to Targeting the Maxillary Nerve
The maxillary nerve can be blocked via intraoral or extraoral approaches. The intraoral approach7 targets the greater palatine foramen but poses risks, such as trigeminal nerve trauma, diplopia, ptosis, and ocular complications, limiting its widespread use by anesthesiologists. Extraoral approaches include the suprazygomatic and infrazygomatic routes. Infrazygomatic approaches, subdivided into anterior8 and posterior,9 carry higher risks of neurovascular injury and are technically more challenging.
The suprazygomatic approach, involving needle entry above the zygomatic arch at the frontozygomatic angle, is favored for safety and consistent anatomical access.10,11 In this approach, the needle's path adjacent to the PPF within the infratemporal fossa involves traversing the following anatomical layers (Figure 6):11
- Subcutaneous fat and fascia
- The superficial layer and deep layer of the temporalis muscle, the fat pad between the muscle layers, and
- The lateral pterygoid muscle and its surrounding fascial coverings.
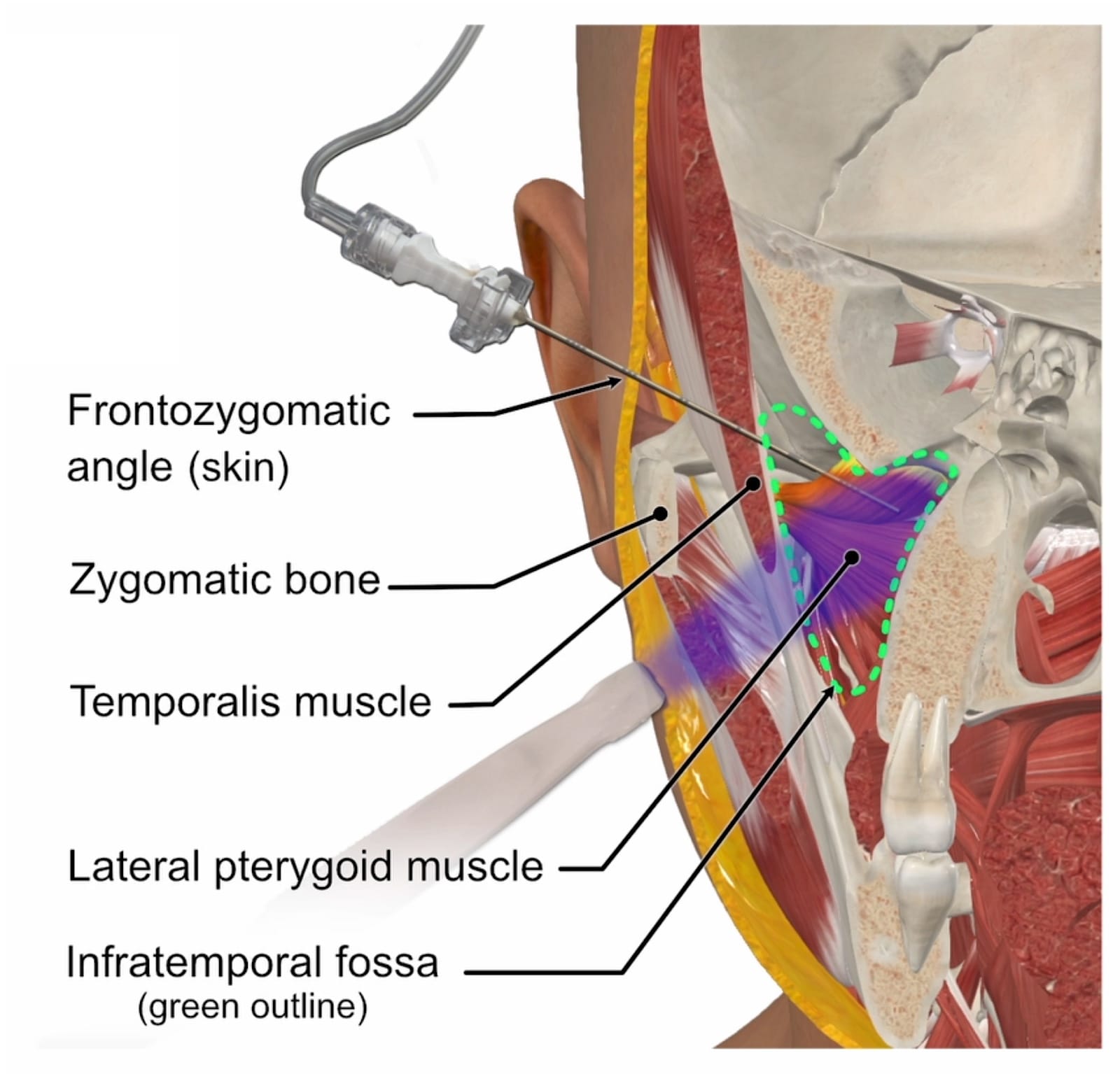
The base image is from CompleteAnatomy.com.
Patient Preparation and Equipment
These blocks are invariably performed under general anesthesia in the pediatric population. Standard ASA monitoring is applied, and sedation or anxiolysis is administered as needed (Figure 7). Ensure the airway is secure and position the head so that the block site is accessible. Skin preparation is performed with an antiseptic solution, such as ophthalmic povidone-iodine or an alcohol wipe (avoiding chlorhexidine due to the risk of ocular and ototoxicity). Ensure the eyes are well-taped; take care to prevent antiseptic solution from spilling into the eyes.
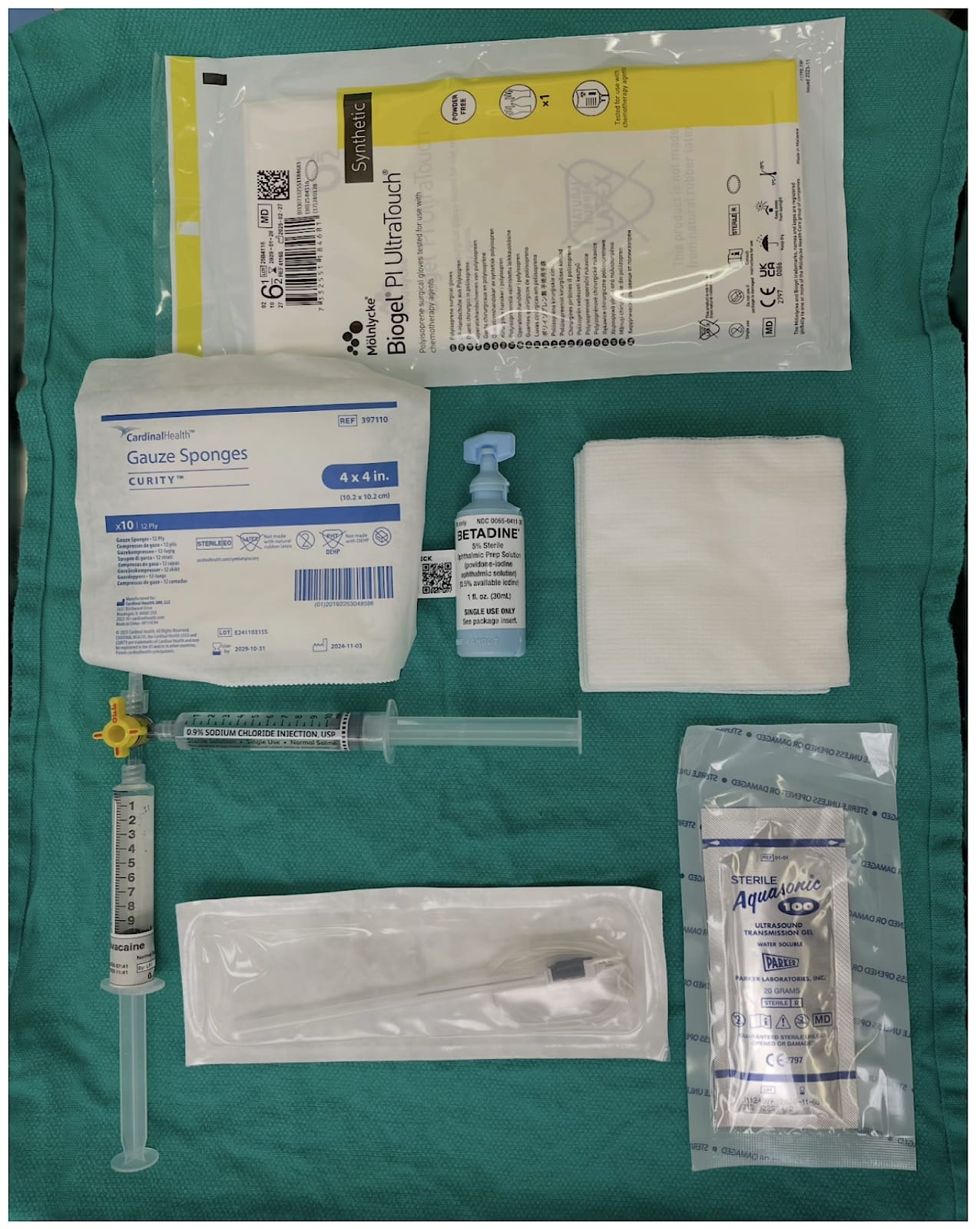
Equipment includes a high-frequency linear ultrasound transducer, a 22–27 ga, 50 mm or smaller echogenic needle, local anesthetics such as ropivacaine or bupivacaine at 0.2-0.5% concentration, and optional adjuncts. Ultrasound gel, sterile covers, syringes, and aspiration devices are also required.
Procedure
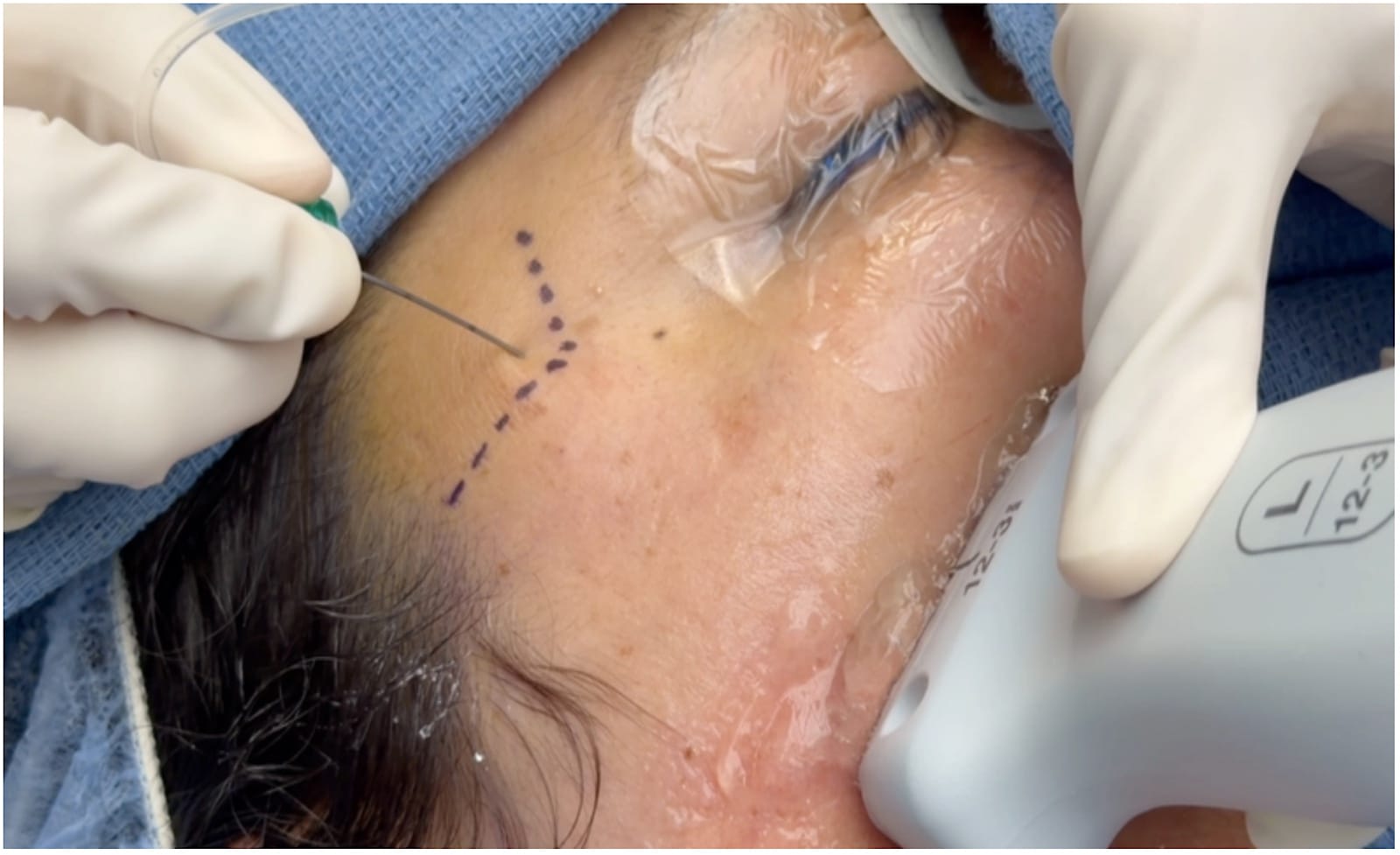
Palpate and mark the frontozygomatic angle located at the confluence of the superior edge of the zygomatic arch and the lateral orbital rim. A 22-27-gauge echogenic needle is inserted perpendicular to the skin at the frontozygomatic angle, advancing carefully until contact with the greater wing of the sphenoid bone is made at around 20 mm (Figure 8).11
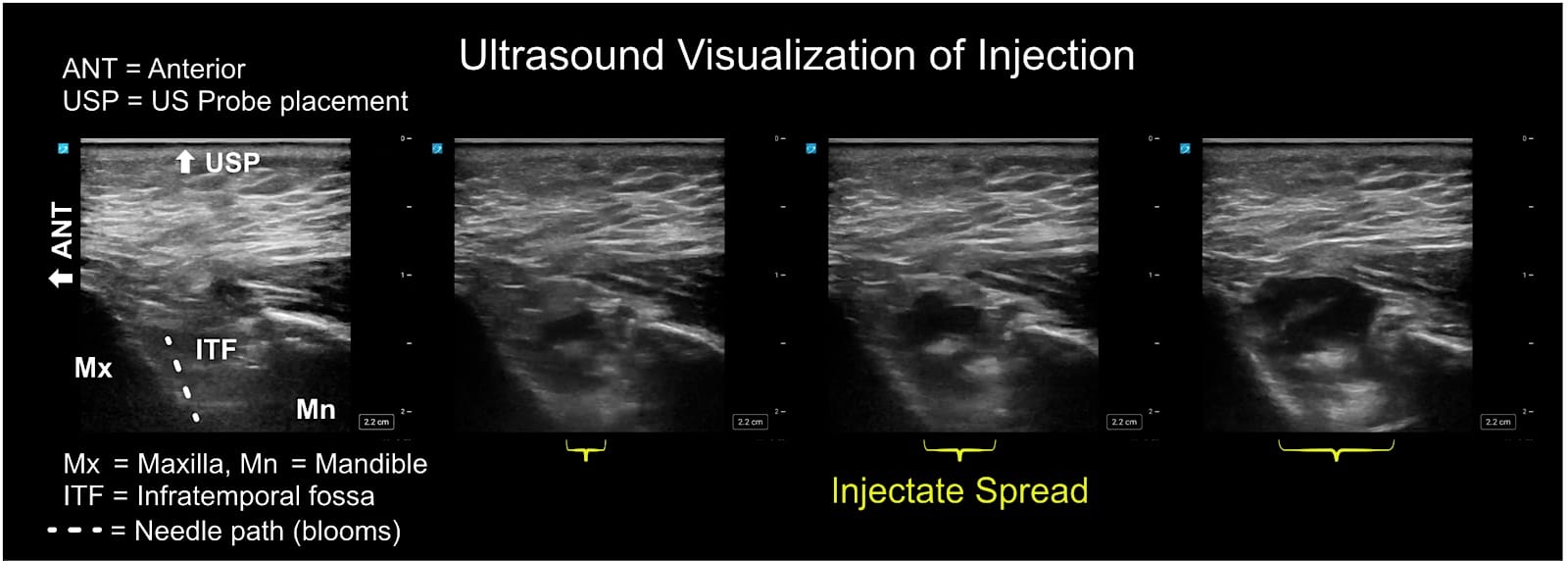
Place a small linear ultrasound probe just inferior to the zygomatic arch, angled approximately 30-45 degrees relative to the transverse plane, and identify landmarks:12 maxilla (Mx), coronoid process of mandible (CPM), and ITF, and infratemporal fossa (Figure 9). The needle is then redirected approximately 5-10 degrees caudad and 10-15 degrees posterior and advanced out of plane under ultrasound guidance. The depth of needle advancement can vary from 20 mm in neonates to 40-50 mm in adults.11,13,14 Confirm the needle position sonographically with saline blooms, aspirate repeatedly, and inject local anesthetic slowly, typically 0.15 ml/kg of 0.15% ropivacaine or bupivacaine, up to a maximum of 5 ml per side), with adjustments for patient size and procedure (Figures 10 and 11).
/2hidi_figure-9.png?sfvrsn=83f9c251_1) Figure 9. Ultrasound display with the maxilla, sphenoid, coronoid process of the mandible, and infratemporal fossa, and the Lateral Pterygoid plate (LPP) labeled.
Figure 9. Ultrasound display with the maxilla, sphenoid, coronoid process of the mandible, and infratemporal fossa, and the Lateral Pterygoid plate (LPP) labeled.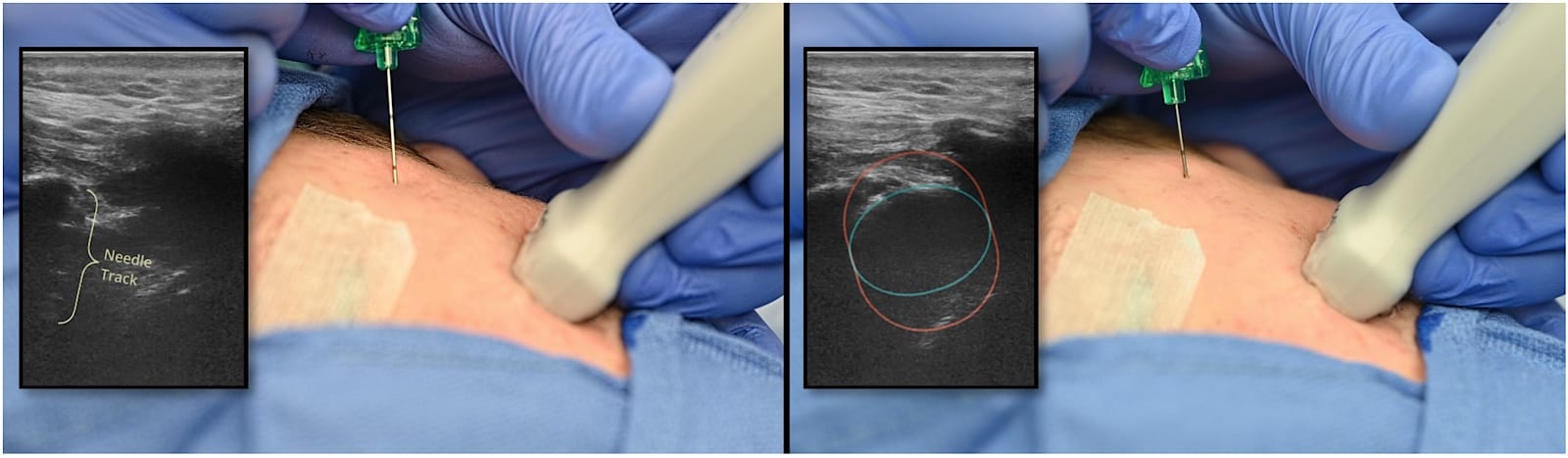
(Insert Video - to come).
Although studies describe slightly different redirection angles,4,11,13,15 it is important to recognize that the SZM nerve block functions like a field block: Injecting in the ITF near the pterygomaxillary fissure enables LA to pass into the PPF, so spread occurs even if the tip is not exactly at the pterygomaxillary fissure or within the fossa itself. If the initial skin puncture is placed posterior to the true frontozygomatic angle, the posterior redirection is reduced, and the final trajectory can be neutral or slightly anterior. There can be inter‑operator variation when the frontozygomatic angle is imprecisely identified. Still, the block can remain effective as a local anesthetic can traverse from the infratemporal fossa into the pterygopalatine fossa, given sufficient volume is used and it is close enough to the pterygomaxillary fissure. However, when excessive local anesthetic is injected, the limited capacity of the pterygopalatine fossa can be exceeded, allowing solution to spill through adjacent foramina into the nasal cavity or orbit, potentially producing unwanted effects from local anesthetic entering via its various communications.
Indications
SZM nerve block can be used to provide analgesia for procedures involving skin and subcutaneous tissue of the midface (CN-V2 distribution); maxilla; palate (entire); and maxillary dentition. In cleft palate repair, Wijnants et al. showed that SZM significantly reduces postoperative opioid requirements and supports early recovery.16 Nores et al. reported lower narcotic use, improved pain scores, and shorter hospitalization with SZM blocks for LeForte procedures.17 For adenotonsillectomies, Lin et al. in their study found markedly reduced opioid consumption, more opioid‑free recoveries, and better early pain control in children receiving the block.1 In adults, SZM has also been used to manage conditions such as trigeminal neuralgia involving CN-V2, post-dural puncture headache, and a variety of other indications.
Contraindications
Absolute contraindications include lack of consent, local infection at the injection site, allergy to local anesthetics, coagulopathy, and distorted facial anatomy that precludes safe needle placement.
Complications and Safety
Reported complications after SZM nerve block are rare but can include transient diplopia, ptosis, strabismus due to local anesthetic diffusion affecting ocular muscles,18 and rarely, facial nerve palsy.7 Symptoms typically resolve within hours without lasting sequelae. Complications related to this block are mainly due to faulty technique and a lack of anatomical understanding. These problems may lead to inadvertent puncture of vascular and neural structures. Use of ultrasound guidance, limiting anesthetic volume, slow incremental injections, and repeated intermittent aspiration minimizes these risks. Injecting too much fluid or using a significant amount of pressure to perform the injection has the potential of causing tissue injury since the PPF is surrounded by bone on multiple sides. Vascular puncture and hematoma formation are possible but uncommon with careful technique. Doppler imaging helps identify vascular structures.
Summary and Conclusions
Apprehension and reluctance about using this block can prevent many patients from benefiting from it. Understanding the anatomy of the ITF and PPF regions can significantly reduce the concern associated with this block. The technique uses an out-of-plane ultrasound approach to inject local anesthetic into a “field” rather than directly adjacent to a specific target.
The SZM nerve block is a safe and effective regional anesthesia technique in pediatric practice. Mastery of relevant sonanatomy, combined with ultrasound guidance, significantly enhances the quality of perioperative analgesia, reduces opioid requirements, and improves postoperative recovery in mid-face surgeries. Proper dosing and vigilance for complications ensure safe implementation (Figure 12).
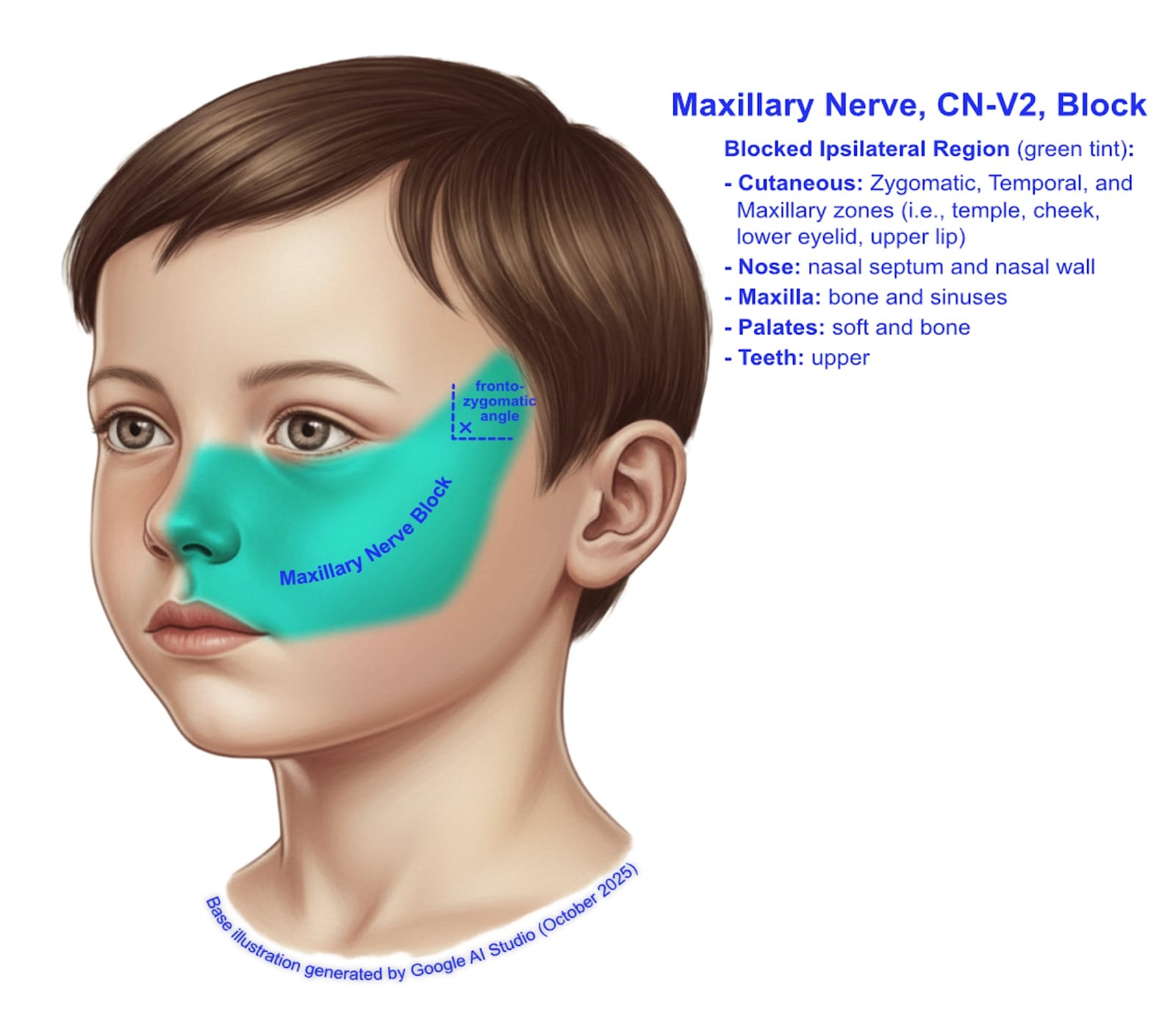
Figure 12. General coverage of maxillary nerve block.





References
- Lin C, Abboud S, Zoghbi V, et al. Suprazygomatic maxillary nerve blocks and opioid requirements in pediatric adenotonsillectomy: a randomized clinical trial. JAMA Otolaryngol Neck Surg2024;150(7):564. https://doi.org/10.1001/jamaoto.2024.1011
- Bergmann I, Heetfeld M, Crozier T, et al. Peripheral nerve blocks give greater hemodynamic stability than general anesthesia for ASA III patients undergoing outpatient knee arthroscopy. Open Med. 2013;8(4):436-42. https://doi.org/10.2478/s11536-012-0143-4
- Shafique S, Das JM. Anatomy, head and neck, maxillary nerve. In: StatPearls. StatPearls Publishing; 2026. https://www.ncbi.nlm.nih.gov/books/NBK542277
- Echaniz G, Chan V, Maynes JT, et al. Ultrasound-guided maxillary nerve block: an anatomical study using the suprazygomatic approach. Can J Anesth Can Anesth 2020;67(2):186-93.https://doi.org/10.1007/s12630-019-01481-x
- Cappello ZJ, Arbor TC, Potts KL. Anatomy, pterygopalatine fossa. In: StatPearls. StatPearls Publishing; 2025. Accessed December 10, 2025. http://www.ncbi.nlm.nih.gov/books/NBK513269
- Sah A, S TG, H CS. Imaging of the infratemporal fossa: a comprehensive pictorial essay. Indian J Surg Oncol 2022;13(4):868-75. https://doi.org/10.1007/s13193-022-01591-x
- Aoun G, Zaarour I, Sokhn S, et al. Maxillary nerve block via the greater palatine canal: An old technique revisited. J Int Soc Prev Community Dent 2015;5(5):359. https://doi.org/10.4103/2231-0762.165930
- Takahashi H, Suzuki T. A novel approach for performing ultrasound-guided maxillary nerve block. J Clin Anesth 2017;43:61-2. https://doi.org/10.1016/j.jclinane.2017.09.015
- Nader A, Kendall MC, De Oliveria GS, et al. Ultrasound-guided trigeminal nerve block via the pterygopalatine fossa: an effective treatment for trigeminal neuralgia and atypical facial pain. Pain Physician 2013;16(5):E537-45.
- Jerman A, Umek N, Cvetko E, et al. Comparison of the feasibility and safety of infrazygomatic and suprazygomatic approaches to pterygopalatine fossa using virtual reality. Reg Anesth Pain Med2023;48(7):359-64. https://doi.org/10.1136/rapm-2022-104068
- Prigge L, Van Schoor A, Bosman MC, et al. Clinical anatomy of the maxillary nerve block in pediatric patients. Lonnqvist P, ed. Pediatr Anesth 2014;24(11):1120-6. https://doi.org/10.1111/pan.12480
- Mireault D, Cawthorn TR, Todd AR, et al. Suprazygomatic maxillary nerve block: an ultrasound and cadaveric study to identify correct sonoanatomical landmarks. J Anesth 2021;35(1):150-3. https://doi.org/10.1007/s00540-020-02877-6
- Marston AP, Merritt G, Morris JM, et al. Impact of age on the anatomy of the pediatric pterygopalatine fossa and its relationship to the suprazygomatic maxillary nerve block. Int J Pediatr Otorhinolaryngol2018;105:85-9. https://doi.org/10.1016/j.ijporl.2017.12.012
- Sola C, Raux O, Savath L, et al. Ultrasound guidance characteristics and efficiency of suprazygomatic maxillary nerve blocks in infants: a descriptive prospective study. Pediatr Anesth 2012;22(9):841-6. https://doi.org/10.1111/j.1460-9592.2012.03861.x
- Peters JJ, Jacobs K, Munill M, et al. The maxillary nerve block in cleft palate care: a review of the literature and experts’ opinion on the preferred technique of administration. J Craniofac Surg2024;35(5):1356-63. https://doi.org/10.1097/SCS.0000000000010343
- Wijnants N, Kappen IFPM, Van Kuijk SMJ, et al. Effectiveness of suprazygomatic maxillary nerve block on postoperative opioid use in pediatric primary cleft palate surgery: a systematic review of the literature. J Plast Reconstr Aesthet Surg 2025;106:174-86. https://doi.org/10.1016/j.bjps.2025.04.046
- Nores GDG, Cuzzone DA, Hush SE, et al. The impact of bilateral suprazygomatic maxillary nerve blocks on postoperative pain control in patients undergoing orthognathic surgery. FACE 2020;1(1):58-65. https://doi.org/10.1177/2732501620949188
- Sved AM, Wong JD, Donkor P, et al. Complications associated with maxillary nerve block anaesthesia via the greater palatine canal. Aust Dent J 1992;37(5):340-5. https://doi.org/10.1111/j.1834-7819.1992.tb00758.x
