Decoding Epidural Maintenance Regimens: Volume, Rate, and Timing for Optimal Labor Analgesia
Cite as: Sjaus A. Decoding epidural maintenance regimens: volume, rate, and timing for optimal labor analgesia. ASRA Pain Medicine News 2026;51. https://doi.org/10.52211/asra050126.014.
Epidural analgesia is the cornerstone of modern obstetric anesthesiologists’ practice. It has evolved through successive advances in neuraxial pharmacology and infusion technology, with recent innovations that leverage the physics of epidural drug delivery.
In 1921, Spanish surgeon Fidel Pagés introduced the concept of single-injection lumbar epidural anesthesia.1 Repeated dosing was made possible by the advent of the epidural catheter in the 1940s. In the 1970s, infusion pumps enabled continuous delivery of local anesthetics (LA).2 However, concentrated LA solutions used at the time often produced dense motor blockade, adversely affecting patient satisfaction and labor progression. Further refinement came through investigations into the impact of LA concentration and dose across a variety of clinical settings.3-5 Through the addition of adjuncts such as lipophilic opioids, these studies allowed the reduction of LA concentration while maintaining analgesic efficacy. Research interests increasingly shifted to epidural side effects (eg, hypotension, pruritus), maternal sense of control during labor, physiological stress responses, and the psychological impact of unrelieved pain. Despite the advancements in continuous epidural infusion (CEA), fluctuating pain intensity during labor required frequent interventions. This amplified anesthesiologists’ workload, setting patient comfort against operational efficiency.
The Return of Bolus-Based Maintenance
The 1980s saw the development of patient-controlled epidural analgesia (PCEA), which allowed patients to administer their own anesthetic boluses, sometimes combined with a background infusion. This method provided greater autonomy, overall satisfaction, and responsiveness to pain. A meta-analysis comparing CEA and PCEA reported that PCEA produced less motor block and required less LA and fewer clinician interventions.6 The differences highlighted the influence of timing, volume, and rate of epidural infusion. As the primary site of action of LA is the epidural nerve roots, with a smaller contribution from subarachnoid diffusion and direct spinal cord effects (opposite for lipophilic opioids), more complete analgesia would be expected from methods that produce greater rostro-caudal spread. In vitro and porcine spine models demonstrated that bolus injections achieved a more extensive spread of LA compared to continuous infusions.7,8
Programmed intermittent epidural bolus (PIEB) was first investigated in two small trials, followed by the landmark trial by Wong et al in 2006.9 By 2018, a Cochrane review found moderate-certainty evidence that PIEB is similar to CEI for safety outcomes and may decrease LA volumes, reduce the risk of breakthrough pain, and improve maternal satisfaction.10 The 2023 update confirmed these findings and reported no association with adverse outcomes, operative delivery, or low Apgar scores.11 A network meta-analysis assessed the effects of CEI, PCEA, PIEB, and their combinations; The PIEB/PCEA combination was superior in primary (maternal satisfaction, rescue analgesia) and secondary outcomes (pain scores, motor block, likelihood of spontaneous delivery, LA consumption).12 Despite the low strength of evidence, these findings pointed to bolus-based techniques for further refinement of epidural labor analgesia.
Infusion Rate and Volume
Garden irrigation provides a useful analogy for understanding how the physics of drug delivery influences the spread of LA. (Figure 1) In a porcine model, infusion of 10 ml of radio-opaque solution using three delivery modalities (continuous drip, a series of small “squirts,” and one “big spray”) showed that the single bolus had significantly greater rostro-caudal spread.13 However, rather than a cohesive advancing front, LA epidural spread is nonuniform, occurring along paths of least resistance in the form of “rivulets” along the nerve root sheath. The most uniform spread occurs with large-volume, high-flow-rate injections under pressure.14 Manual injection can generate flow rates up to 1200 mL/h, whereas specialized PIEB pumps are capable of >300 mL/h, compared with 8-12 mL/h for CEI. Flow characteristics are further influenced by in-line filters, tubing dimensions and syringe size.15,16 However, the gains in pain outcomes and segmental spread seem to plateau at a PIEB rate of around 100 ml/h.17,18
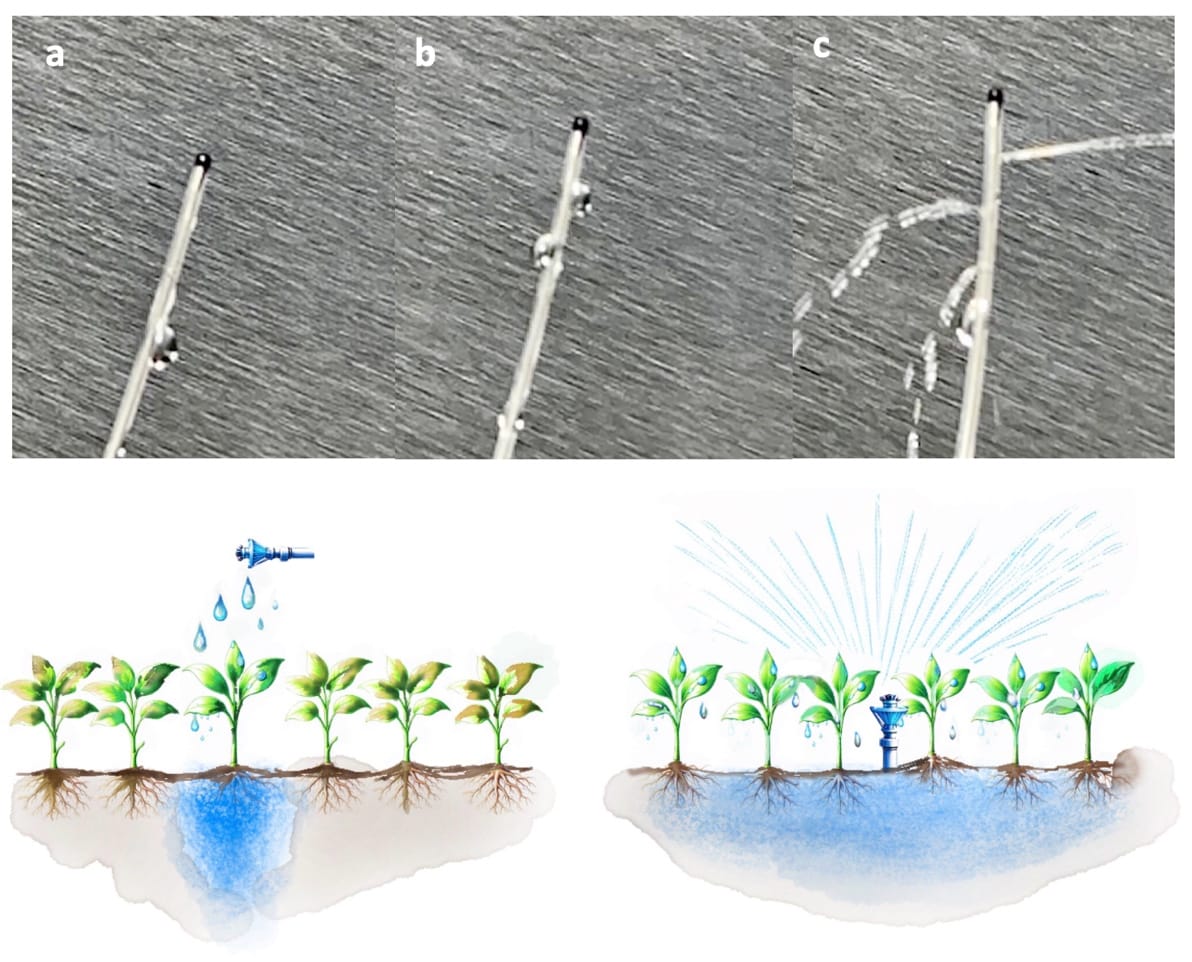
A PIEB volume of 10 mL delivered at regular intervals achieved effective analgesia in 90% of patients (EV90), with low rates of breakthrough pain and motor block.19 Larger boluses (eg, 12-15 mL) did not result in substantially improved efficacy and were associated with increased motor block. Conversely, progressively decreasing bolus volumes increased the incidence of breakthrough pain.20
Catheter design also affects LA distribution; the number, size, and position of orifices influence both the flow rate and the direction of spread. At low infusion pressures, flow occurs primarily through proximal orifices. Therefore, higher pressures and flow rates enhance the effectiveness of multi-orifice catheters in distributing LA.7,21 However, smaller gauge catheters (20G) will trigger frequent occlusion alarms at high infusion rates.15
In conclusion, while all epidural infusion regimens offer safe and effective labor analgesia, the combined PIEB/PCEA leverages the physics and timing of epidural injection to optimize the spread of local anesthetic, providing effective analgesia in response to fluctuating labor pain.
The Importance of Intervals
The PIEB interval seems directly proportional to bolus volume, with a range of 35 to 45 minutes for 8-10 mL boluses.22-24 Shorter intervals increase LA consumption, while longer intervals increase the incidence of breakthrough pain.23, 25-26
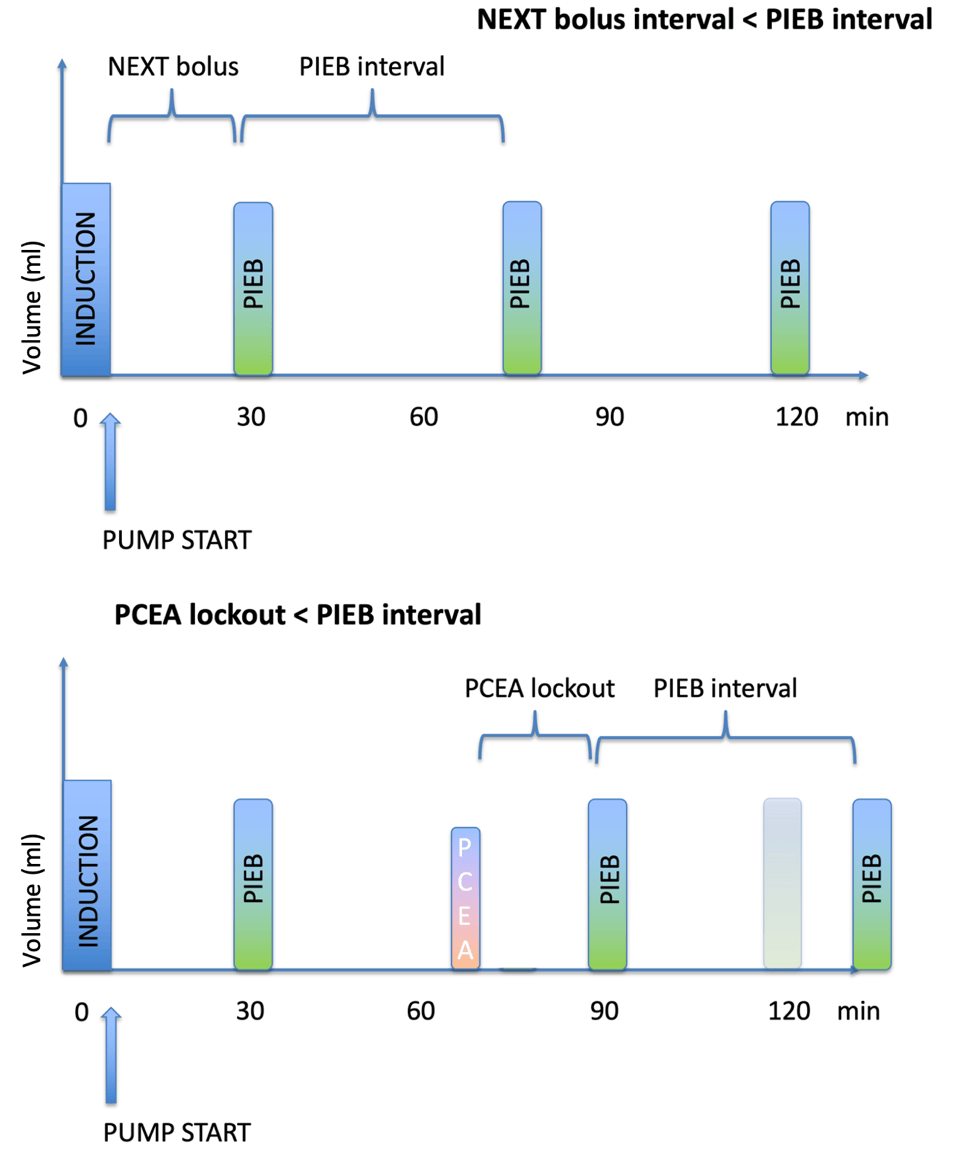
Additional interval-related considerations arise when PIEB is combined with PCEA; the interval immediately following the induction dose is termed the next bolus (PIEBnb), while the time between PCEA doses and subsequent PIEB is the lockout interval. (Figure 2). These interdependent parameters with complex, non-linear relationships require an innovative approach. Munro et al. used specialized mathematical modeling to optimize maternal satisfaction, clinician workload, and PCEA/PIEB ratio, estimating PIEBnb at 29.4 minutes, PIEB interval at 59.8 minutes, and PIEB volume at 6.2 mL (ropivacaine 0.1% with 2 µg/mL fentanyl).27Although the findings somewhat diverge from the standard settings currently used at our institution, they uphold the rule-of-thumb that all other interval settings should be shorter compared to the PIEB interval, as PIEB volume is typically set larger than PCEA volume. (Figure 2)
Effects Beyond Analgesia
Beyond analgesia and satisfaction, PIEB may improve clinician workload, shorten the second stage of labor, and reduce the incidence of forceps delivery.28-31 However, a randomized trial involving 132 participants found no significant differences in labor duration, total drug consumption, maternal satisfaction, sensory and motor block levels, presence of unilateral motor block, hemodynamics, side effects, mode of delivery, or newborn outcomes.32 Two studies found a lower risk of peri-partum maternal fever despite a similar inflammatory marker profile and no difference in temperature in another study.33-35 Lower LA consumption (per hour and cumulative dose) may reduce overall systemic accumulation and the risk of LA toxicity.12 While these findings are reassuring for labor progress and neonatal outcomes, further studies are needed in high-risk populations to explore the combined effects with different epidural techniques (combined spinal-epidural, dural puncture epidural) and pharmacological adjuncts, as well as continued refinement of volume and interval parameters.
Limitations of Bolus-Based Infusions
Despite the evidence for superior analgesic coverage and fewer side effects, the effect sizes for bolus techniques are modest and maternal satisfaction with any neuraxial technique is high. The potential safety concerns of PIEB include higher sensory block levels despite less LA being used, the risk of catheter migration due to the “pressurization effect,” and the need for standardization to avoid errors with multi-parameter settings, while avoiding the “one size fits all” approach.18,36 Although the additional cost of technology (PIEB capable pumps) may limit widespread adoption, operational benefits may justify the investment.37
In conclusion, while all epidural infusion regimens offer safe and effective labor analgesia, the combined PIEB/PCEA leverages the physics and timing of epidural injection to optimize the spread of local anesthetic, providing effective analgesia in response to fluctuating labor pain. The current evidence suggests that, compared with CEA or CEA/PCEA, PIEB/PCEA improves analgesic efficacy and maternal satisfaction while reducing side effects.

References
- Capogna G. History of lumbar epidural block. In: Epidural Technique In Obstetric Anesthesia. Springer International Publishing; 2020:1-18. https://doi.org/10.1007/978-3-030-45332-9_1
- Brill S, Gurman GM, Fisher A. A history of neuraxial administration of local analgesics and opioids. Eur J Anaesthesiol 2005;20(9):682-9. https://doi.org/10.1017/S026502150300111X
- Scott DA, Chamley DM, Mooney PH, et al. Epidural ropivacaine infusion for postoperative analgesia after major lower abdominal surgery–a dose finding study. Anesth Analg1995;81(5):982-6. https://doi.org/10.1097/00000539-199511000-00015
- Ngan Kee WD, Ng FF, Khaw KS, et al. Determination and comparison of graded dose-response curves for epidural bupivacaine and ropivacaine for analgesia in laboring nulliparous women. Anesthesiology 2010;113(2):445-53. https://doi.org/10.1097/ALN.0b013e3181bdf9da
- Curatolo M, Schnider TW, Petersen-Felix S, et al. A direct search procedure to optimize combinations of epidural bupivacaine, fentanyl, and clonidine for postoperative analgesia. Anesthesiology 2000;92(2):325-37. https://doi.org/10.1097/00000542-200002000-00012
- Van Der Vyver M, Halpern S, Joseph G. Patient-controlled epidural analgesia versus continuous infusion for labour analgesia: a meta-analysis. Br J Anaesth 2002;89(3):459-65. https://doi.org/10.1093/bja/89.3.459
- Kaynar AM, Shankar KB. Epidural infusion: continuous or bolus? Anesth Analg 1999;89(2):534. https://doi.org/10.1097/00000539-199908000-00063
- Mowat I, Tang R, Vaghadia H, et al. Epidural distribution of dye administered via an epidural catheter in a porcine model. Br J Anaesth 2016;116(2):277-81. https://doi.org/10.1093/bja/aev432
- Wong CA, Ratliff JT, Sullivan JT, et al. A randomized comparison of programmed intermittent epidural bolus with continuous epidural infusion for labor analgesia. Anesth Analg 2006;102(3):904-9. https://doi.org/10.1213/01.ane.0000197778.57615.1a
- Sng BL, Zeng Y, De Souza NNA, et al. Automated mandatory bolus versus basal infusion for maintenance of epidural analgesia in labour. Cochrane Anaesthesia Group, ed. Cochrane Database of Systematic Reviews 2018;2018(5). https://doi.org/10.1002/14651858.CD011344.pub2
- Tan HS, Zeng Y, Qi Y, et al. Automated mandatory bolus versus basal infusion for maintenance of epidural analgesia in labour. Cochrane Anaesthesia Group, ed. Cochrane Database of Systematic Reviews 2023;2023(6). https://doi.org/10.1002/14651858.CD011344.pub3
- Wydall S, Zolger D, Owolabi A, et al. Comparison of different delivery modalities of epidural analgesia and intravenous analgesia in labour: a systematic review and network meta-analysis. Can J Anesth/J Can Anesth 2023;70(3):406-42. https://doi.org/10.1007/s12630-022-02389-9
- Cole J, Hughey S. Bolus epidural infusion improves spread compared with continuous infusion in a cadaveric porcine spine model. Reg Anesth Pain Med 2019;44(12):1080-3. https://doi.org/10.1136/rapm-2019-100818
- Hogan Q. Distribution of solution in the epidural space: Examination by cryomicrotome section. Reg Anesth Pain Med 2002;27(2):150-6. https://doi.org/10.1053/rapm.2002.29748
- Krawczyk P, Piwowar P, Sałapa K, et al. Do epidural catheter size and flow rate affect bolus injection pressure in different programmed intermittent epidural bolus regimens? an in vitro study. Anesth Analg 2019;129(6):1587-94. https://doi.org/10.1213/ANE.0000000000003650
- Younger JD, Faryami A, Prasad M, et al. Direct comparison of peak bulk flow rate of programmable intermittent epidural bolus and manual epidural bolus using a closed-end multiorifice catheter: an experimental study. Anesth Analg 2023;136(6):1198-1205. https://doi.org/10.1213/ANE.0000000000006268
- Lange EMS, Wong CA, Fitzgerald PC, et al. Effect of epidural infusion bolus delivery rate on the duration of labor analgesia: a randomized clinical trial. Anesthesiology 2018;128(4):745-53. https://doi.org/10.1097/ALN.0000000000002089
- Casellato JF, Ye XY, Downey K, et al. Changes in sensory block level during a programmed intermittent epidural bolus regimen for labour analgesia: a prospective observational cohort study. Can J Anesth/J Can Anesth 2022;69(12):1471-6. https://doi.org/10.1007/s12630-022-02318-w
- Zakus P, Arzola C, Bittencourt R, et al. Determination of the optimal programmed intermittent epidural bolus volume of bupivacaine 0.0625% with fentanyl 2 μg.ml−1 at a fixed interval of forty minutes: a biased coin up‐and‐down sequential allocation trial. Anaesthesia 2018;73(4):459-65. https://doi.org/10.1111/anae.14159
- Zuo RH, Dang JJ, Zhuang JW, et al. The incidence of breakthrough pain associated with programmed intermittent bolus volumes for labor epidural analgesia: a randomized controlled trial. Int J Obstet Anesth 2022;51:103571. https://doi.org/10.1016/j.ijoa.2022.103571
- Power I, Thorburn J. Differential flow from multihole epidural catheters. Anaesthesia1988;43(10):876-8. https://doi.org/10.1111/j.1365-2044.1988.tb05605.x
- Epsztein Kanczuk M, Barrett NM, Arzola C, et al. Programmed intermittent epidural bolus for labor analgesia during first stage of labor: a biased-coin up-and-down sequential allocation trial to determine the optimum interval time between boluses of a fixed volume of 10 mL of bupivacaine 0.0625% with fentanyl 2 μg/mL. Anesth Analg 2017;124(2):537-41. https://doi.org/10.1213/ANE.0000000000001655
- Yao HQ, Huang JY, Deng JL, et al. Randomized assessment of the optimal time interval between programmed intermittent epidural boluses when combined with the dural puncture epidural technique for labor analgesia. Anesth Analg 2023;136(3):532-9. https://doi.org/10.1213/ANE.0000000000006201
- Mei Z, Wang Q, Song S, et al. Optimum programmed intermittent epidural bolus interval time of ropivacaine 0.0625% with dexmedetomidine 0.4 μg/ml at a fixed volume of 10 mL: a randomized controlled trial. Front Pharmacol 2024;15:1368222. https://doi.org/10.3389/fphar.2024.1368222
- Bittencourt R, Arzola C, Zakus P, et al. A biased coin up-and-down sequential allocation trial to determine the optimum programmed intermittent epidural bolus time interval between 5 mL boluses of bupivacaine 0.125% with fentanyl 2 µg·mL−1. Can J Anesth/J Can Anesth2019;66(9):1075-81. https://doi.org/10.1007/s12630-019-01407-7
- Shatalin D, Arzola C, Downey K, et al. Programmed intermittent epidural bolus for labour analgesia during first stage of labour: a sequential allocation trial to determine the effective interval time between boluses of a fixed volume of 2.5 mL of bupivacaine 0.25% plus fentanyl 8 µg·mL−1. Can J Anesth/J Can Anesth 2021;68(5):653-60. https://doi.org/10.1007/s12630-021-01922-6
- Munro A, George RB, Andreou P. An innovative approach to determine programmed intermittent epidural bolus pump settings for labor analgesia: a randomized controlled trial. Anesth Analg2024;139(3):545-54. https://doi.org/10.1213/ANE.0000000000006813
- Carvalho B, Cohen SE, Giarrusso K, et al. “Ultra-light” patient-controlled epidural analgesia during labor: effects of varying regimens on analgesia and physician workload. Int J Obstet Anesth2005;14(3):223-9. https://doi.org/10.1016/j.ijoa.2005.02.003
- McKenzie CP, Cobb B, Riley ET, et al. Programmed intermittent epidural boluses for maintenance of labor analgesia: an impact study. Int J Obstet Anesth 2016;26:32-8. https://doi.org/10.1016/j.ijoa.2015.11.005
- Bullingham A, Liang S, Edmonds E, et al. Continuous epidural infusion vs programmed intermittent epidural bolus for labour analgesia: a prospective, controlled, before-and-after cohort study of labour outcomes. Br J Anaesth 2018;121(2):432-7.https://doi.org/10.1016/j.bja.2018.03.038
- Capogna G, Camorcia M, Stirparo S, et al. Programmed Intermittent epidural bolus versus continuous epidural infusion for labor analgesia: the effects on maternal motor function and labor outcome. A Randomized Double-Blind Study in Nulliparous Women. Anesth Analg2011;113(4):826-31. https://doi.org/10.1213/ANE.0b013e31822827b8
- Ferrer LE, Romero DJ, Vásquez OI, et al. Effect of programmed intermittent epidural boluses and continuous epidural infusion on labor analgesia and obstetric outcomes: a randomized controlled trial. Arch Gynecol Obstet 2017;296(5):915-22. https://doi.org/10.1007/s00404-017-4510-x
- Fan Y, Hou W, Feng S, et al. Programmed intermittent epidural bolus decreases the incidence of intra-partum fever for labor analgesia in primiparous women: a randomized controlled study. Arch Gynecol Obstet 2019;300(6):1551-7. https://doi.org/10.1007/s00404-019-05354-y
- Li B, Yuan S, Chen A, et al. Programmed intermittent epidural bolus at different intervals combined with patient-controlled epidural analgesia on body temperature during labour analgesia. J Coll Physicians Surg Pak 2020;30(05):463-6. https://doi.org/10.29271/jcpsp.2020.05.463
- Feng SW, Xu SQ, Ma L, et al. Regular intermittent bolus provides similar incidence of maternal fever compared with continuous infusion during epidural labor analgesia. Saudi Med J2014;35(10):1237-42. https://doi.org/
- Betti F, Carvalho B, Riley ET. Intrathecal migration of an epidural catheter while using a programmed intermittent epidural bolus technique for labor analgesia maintenance: a case report. A & A Case Reports 2017;9(12):357-9. https://doi.org/10.1213/XAA.0000000000000616
- Carvalho B, George RB, Cobb B, et al. Implementation of programmed intermittent epidural bolus for the maintenance of labor analgesia. Anesth Analg 2016;123(4):965-71. https://doi.org/10.1213/ANE.0000000000001407